Aqueous Crystal Growth and Dissolution Kinetics of Earth Surface Materials Active
Although calcium carbonate reaction kinetics has important application in several areas of Earth Science, the mechanism of natural organic matter mediation of carbonate minerals growth and dissolution rates remains largely unknown. This project uses multiple approaches to study calcium carbonate formation and dissolution rates in surface water and groundwater systems.
Background
Project work on calcium carbonate reaction kinetics has important application in several areas of earth science, including water quality concerns and the global carbon budget. The mechanism of natural organic matter mediation of carbonate minerals growth and dissolution rates remains largely unknown. We use laboratory, field, and theoretical approaches to study calcium carbonate formation and dissolution rates in surface water and groundwater systems.
Important aspects of project research include characterization of the interaction of natural organic material with calcium carbonate surfaces during crystal growth and dissolution. We develop models to evaluate natural organic material influence on calcium carbonate crystal growth and dissolution kinetics. Other project activities focus on the development and verification of methods to characterize natural organic matter-metal ion speciation.
Project staff sample surface and ground water for chemical and isotopic composition related to calcium carbonate formation and dissolution. We use geochemical mass balance calculations to identify possible calcium carbonate formation and dissolution reactions in a range of surface and ground water settings. Project staff also monitor calcium carbonate crystal growth and dissolution in conjunction with tools developed for dating young groundwaters.
Our project uses techniques of low-temperature geochemistry to interpret and characterize chemical processes, such as precipitation of calcium carbonate in surface lake water (see photos below), occurring in surface water and ground water. We study metal binding by dissolved organic matter and crystal growth and dissolution reaction rate mediation by dissolved constituents because metal ion speciation and mineral formation rates influence water quality and carbon storage. Metal ion complexation with organic matter mediates metal ion reactivity in surface water and ground water -- metal ion speciation controls metal ion chemical and biogeochemical reactivity and bioavailability.
We determine equilibrium metal species distributions in surface and ground water containing organic matter, and characterize crystal growth and dissolution rates in surface and ground water containing organic material.
Project Interests
- Calcium carbonate nucleation and growth in surface and groundwater
- Calcium carbonate formation and dissolution in arid soils at the Amargosa Desert Research Site located near Beatty, Nevada.
Pyramid Lake Research



Aqueous Crystal Growth and Dissolution Kinetics of Calcium Carbonate Minerals at the Amarigosa Desert Research Site, Nevada
Below are publications associated with this project.
Specific surface area of a crushed welded tuff before and after aqueous dissolution
Effects of acid rain and sulfur dioxide on marble dissolution
Welded tuff porosity characterization using mercury intrusion, nitrogen and ethylene glycol monoethyl ether sorption and epifluorescence microscopy
Employment of Gibbs-Donnan-based concepts for interpretation of the properties of linear polyelectrolyte solutions
Modeling electrostatic and heterogeneity effects on proton dissociation from humic substances
Description and testing of three moisture sensors for measuring surface wetness on carbonate building stones
Interpretation with a Donnan-based concept of the influence of simple salt concentration on the apparent binding of divalent ions to the polyelectrolytes polystyrenesulfonate and dextran sulfate
Proton and metal ion binding to natural organic polyelectrolytes-I. Studies with synthetic model compounds
Proton and metal ion binding to natural organic polyelectrolytes-II. Preliminary investigation with a peat and a humic acid
Crystal growth of calcite from calcium bicarbonate solutions at constant PCO2 and 25°C: a test of a calcite dissolution model
Below are software products associated with this project.
- Overview
Although calcium carbonate reaction kinetics has important application in several areas of Earth Science, the mechanism of natural organic matter mediation of carbonate minerals growth and dissolution rates remains largely unknown. This project uses multiple approaches to study calcium carbonate formation and dissolution rates in surface water and groundwater systems.
Background
 Sources/Usage: Public Domain. View Media Details
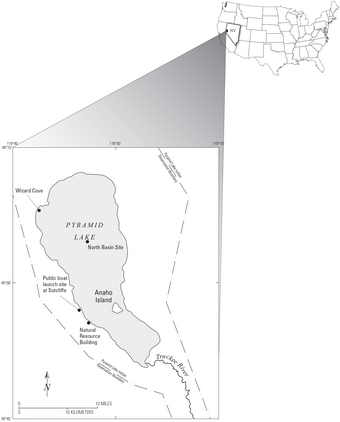
Sources/Usage: Public Domain. View Media DetailsMap showing research site at Pyramid Lake, NV, for the Aqueous Crystal Growth and Dissolution Kinetics of Calcium Carbonate Minerals project. Project work on calcium carbonate reaction kinetics has important application in several areas of earth science, including water quality concerns and the global carbon budget. The mechanism of natural organic matter mediation of carbonate minerals growth and dissolution rates remains largely unknown. We use laboratory, field, and theoretical approaches to study calcium carbonate formation and dissolution rates in surface water and groundwater systems.
Important aspects of project research include characterization of the interaction of natural organic material with calcium carbonate surfaces during crystal growth and dissolution. We develop models to evaluate natural organic material influence on calcium carbonate crystal growth and dissolution kinetics. Other project activities focus on the development and verification of methods to characterize natural organic matter-metal ion speciation.
Project staff sample surface and ground water for chemical and isotopic composition related to calcium carbonate formation and dissolution. We use geochemical mass balance calculations to identify possible calcium carbonate formation and dissolution reactions in a range of surface and ground water settings. Project staff also monitor calcium carbonate crystal growth and dissolution in conjunction with tools developed for dating young groundwaters.
Our project uses techniques of low-temperature geochemistry to interpret and characterize chemical processes, such as precipitation of calcium carbonate in surface lake water (see photos below), occurring in surface water and ground water. We study metal binding by dissolved organic matter and crystal growth and dissolution reaction rate mediation by dissolved constituents because metal ion speciation and mineral formation rates influence water quality and carbon storage. Metal ion complexation with organic matter mediates metal ion reactivity in surface water and ground water -- metal ion speciation controls metal ion chemical and biogeochemical reactivity and bioavailability.
We determine equilibrium metal species distributions in surface and ground water containing organic matter, and characterize crystal growth and dissolution rates in surface and ground water containing organic material.
Project Interests
- Calcium carbonate nucleation and growth in surface and groundwater
- Calcium carbonate formation and dissolution in arid soils at the Amargosa Desert Research Site located near Beatty, Nevada.
Pyramid Lake Research
 Sources/Usage: Public Domain. View Media Details
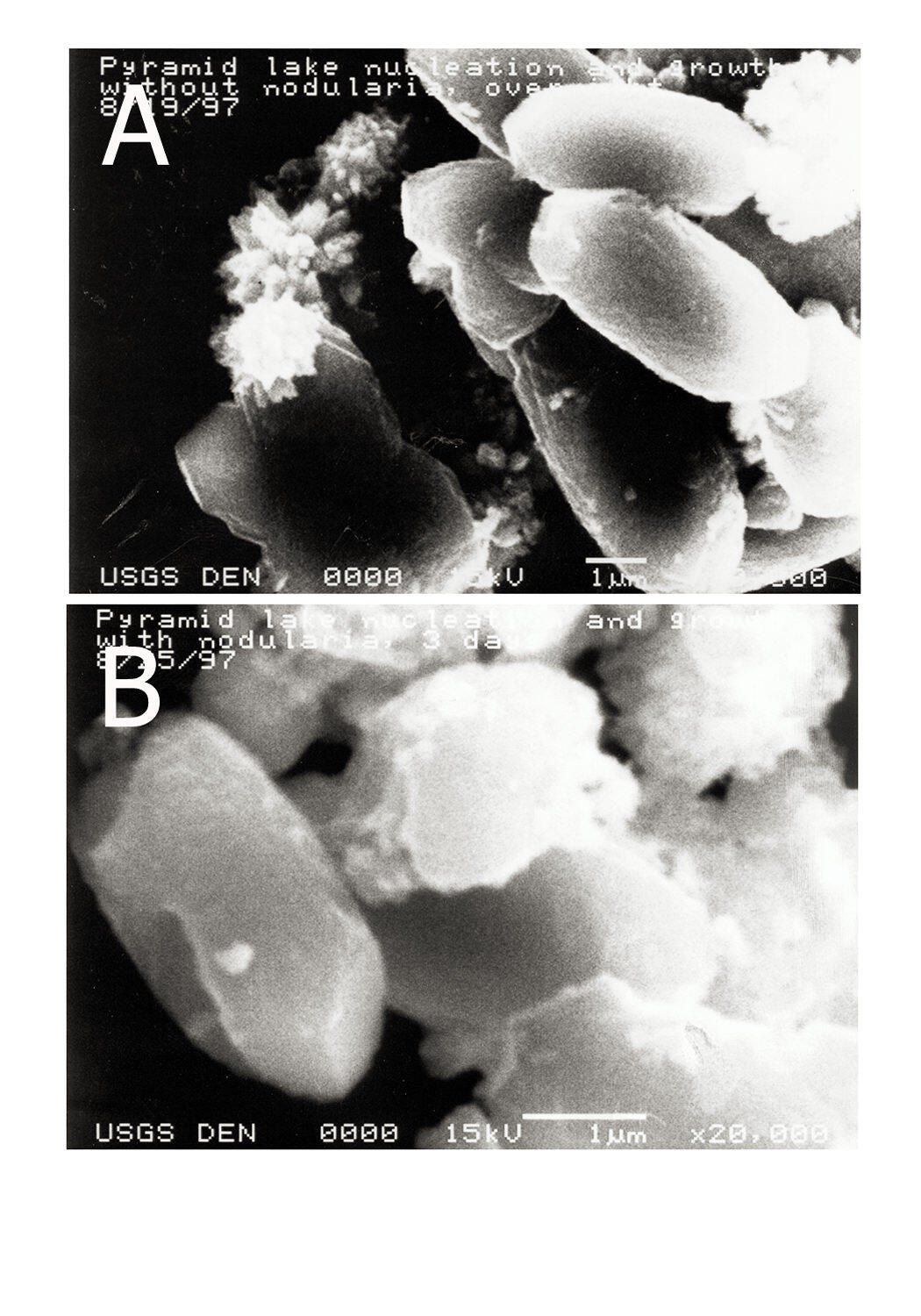
Sources/Usage: Public Domain. View Media DetailsScanning Electron Microscopy (SEM) images of calcium carbonate particles formed in a Pyramid Lake surface-water sample during nucleation experiments (magnification bar shown on each figure). A) Control experiment - nucleation in the absence of algae; B) Calcium carbonate nucleation in a sample containing algae.  Sources/Usage: Public Domain. View Media Details
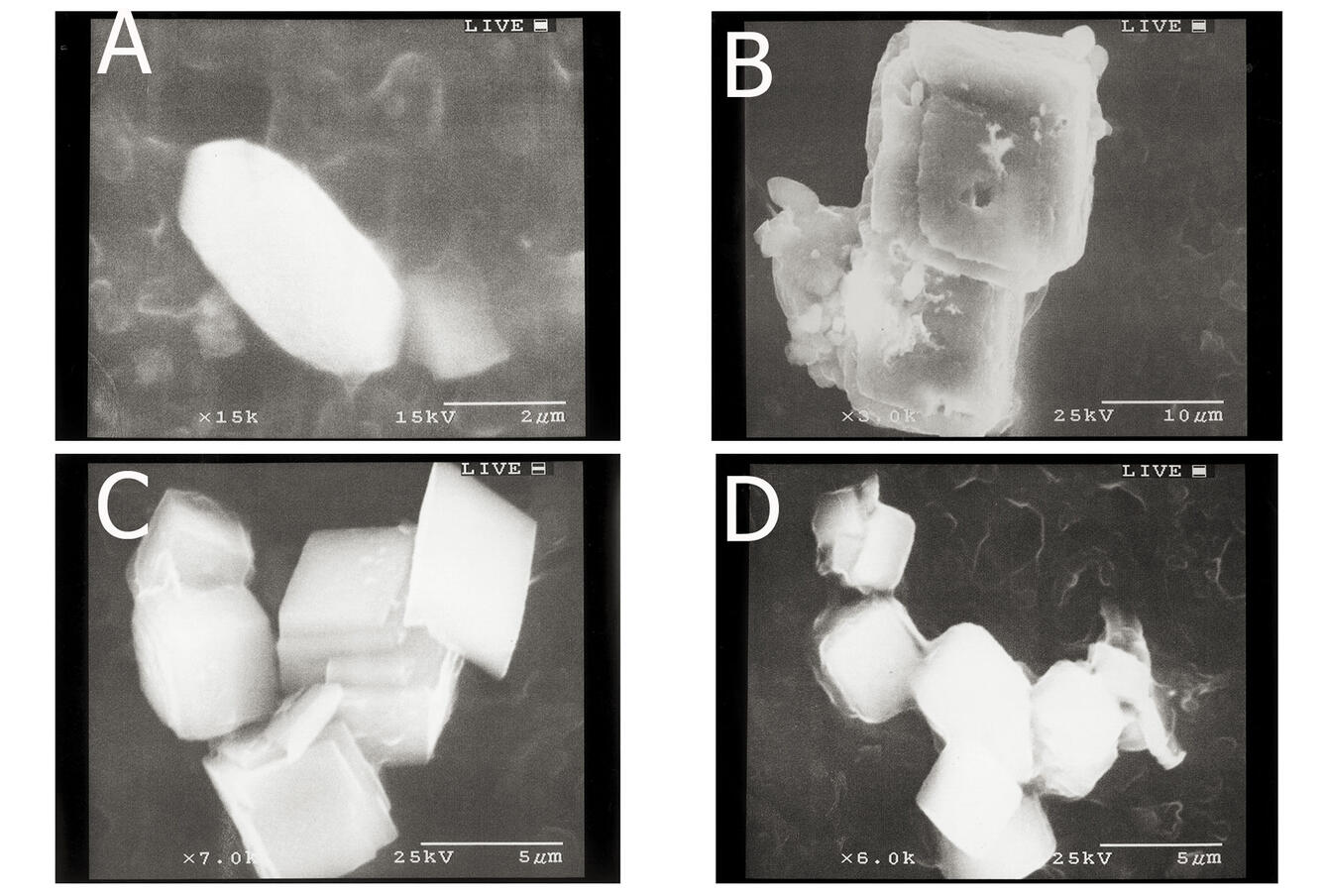
Sources/Usage: Public Domain. View Media DetailsScanning Electron Microscopy (SEM) images of calcium carbonate particles in a (North Basin) Pyramid Lake surface-water sample collected during August 2000. A) aragonite particle, B) calcite particle, C) calcite particle, and D) calcite particle. Scale marker is at the bottom of the photo.  Sources/Usage: Public Domain. View Media Details
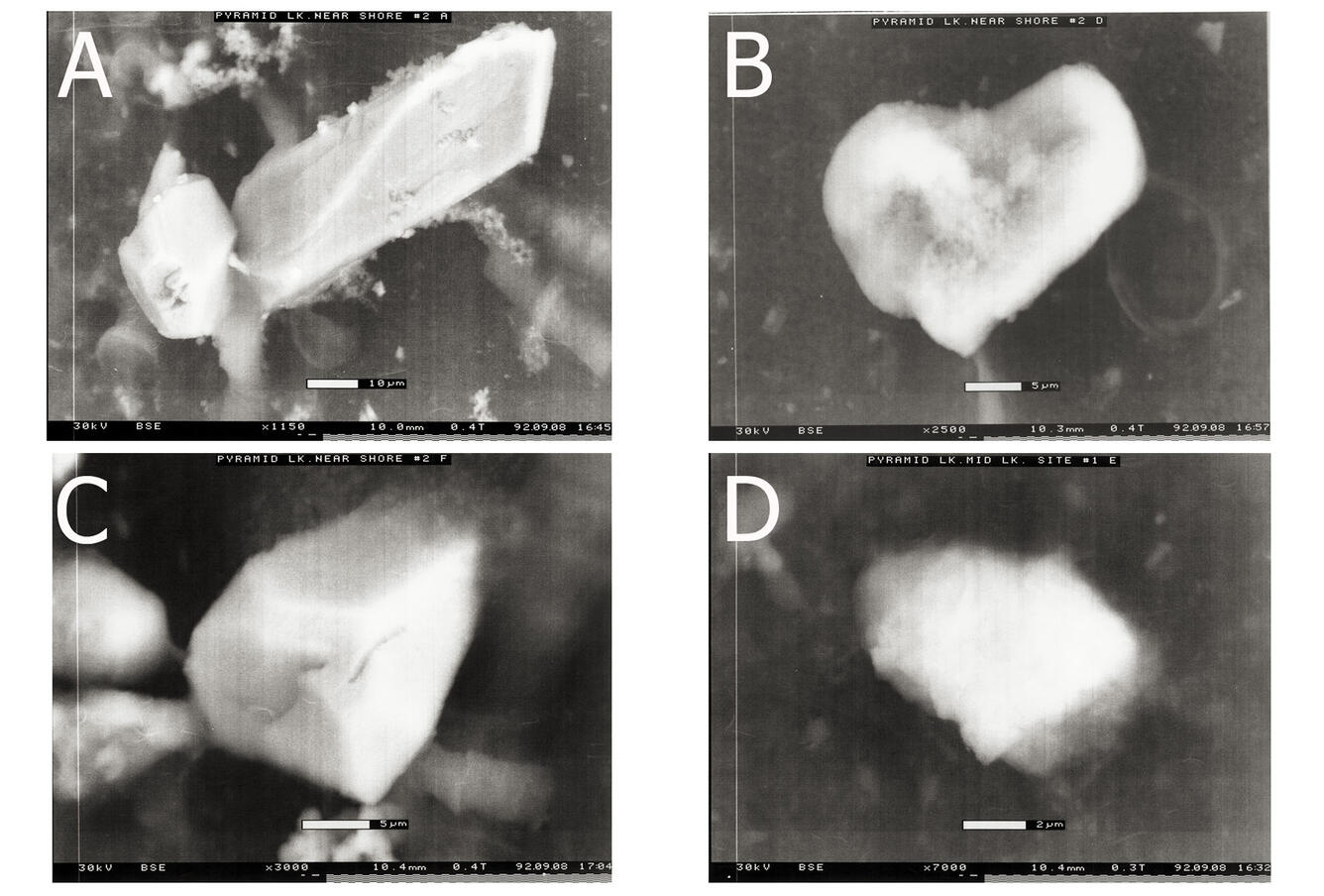
Sources/Usage: Public Domain. View Media DetailsCalcium carbonate particles from Pyramid Lake surface-water samples collected near shore at the Natural Resource Building (August 1992) A), B), and C) and from a mid-lake location D). Scale marker is at the bottom of the photo.  Sources/Usage: Public Domain. View Media Details
Sources/Usage: Public Domain. View Media DetailsView of Pyramid Lake from the shore, taken in 1992. Characteristic large carbonate mounds are on the left and are well above the present lake level. A tree close to the present lake shore gives scale.  Sources/Usage: Public Domain. View Media Details
Sources/Usage: Public Domain. View Media DetailsA cross-section of a carbonate mound along the shore of Pyramid Lake exhibiting the characteristic radial distribution of crystals found near the mound center. The key at the center of the photo is about one inch in diameter (1992).  Sources/Usage: Public Domain. View Media Details
Sources/Usage: Public Domain. View Media DetailsHot Springs at the Needles Area of Pyramid Lake, NV (below) shows active calcium carbonate precipitation. For more information see Reddy and Hoch 2012. - Science
Aqueous Crystal Growth and Dissolution Kinetics of Calcium Carbonate Minerals at the Amarigosa Desert Research Site, Nevada
The USGS studies calcium carbonate mineral reactions in soils at the Amarigosa Desert Research Site (ADRS) near Beatty, Nevada, in order to better understand carbon dioxide transport to and from the soil surface, as well as sequestration of toxic elements in the soil. - Publications
Below are publications associated with this project.
Filter Total Items: 58Specific surface area of a crushed welded tuff before and after aqueous dissolution
Specific surface areas were measured for several reference minerals (anorthoclase, labradorite and augite), welded tuff and stream sediments from Snowshoe Mountain, near Creede, Colorado. Crushed and sieved tuff had an unexpectedly small variation in specific surface area over a range of size fractions. Replicate surface area measurements of the largest and smallest tuff particle size fractions exAuthorsM.M. Reddy, H. C. ClaassenEffects of acid rain and sulfur dioxide on marble dissolution
Acid precipitation and the dry deposition of sulfur dioxide (SO2) accelerate damage to carbonate-stone monuments and building materials. This study identified and quantified environmental damage to a sample of Vermont marble during storms and their preceding dry periods. Results from field experiments indicated the deposition of SO2 gas to the stone surface during dry periods and a twofold increasAuthorsPaul F. Schuster, Michael M. Reddy, Susan I. SherwoodWelded tuff porosity characterization using mercury intrusion, nitrogen and ethylene glycol monoethyl ether sorption and epifluorescence microscopy
Porosity of welded tuff from Snowshoe Mountain, Colorado, was characterized by mercury intrusion porosimetry (MIP), nitrogen sorption porosimetry, ethylene glycol monoethyl ether (EGME) gas phase sorption and epifluorescence optical microscopy. Crushed tuff of two particle-size fractions (1-0.3 mm and less than 0.212 mm), sawed sections of whole rock and crushed tuff that had been reacted with 0.1AuthorsM.M. Reddy, H. C. Claassen, D.W. Rutherford, C. T. ChiouEmployment of Gibbs-Donnan-based concepts for interpretation of the properties of linear polyelectrolyte solutions
Earlier research has shown that the acid dissociation and metal ion complexation equilibria of linear, weak-acid polyelectrolytes and their cross-linked gel analogues are similarly sensitive to the counterion concentration levels of their solutions. Gibbs-Donnan-based concepts, applicable to the gel, are equally applicable to the linear polyelectrolyte for the accommodation of this sensitivity toAuthorsJ.A. Marinsky, M.M. ReddyModeling electrostatic and heterogeneity effects on proton dissociation from humic substances
No abstract available.AuthorsE. Tipping, M.M. Reddy, M.A. HurleyDescription and testing of three moisture sensors for measuring surface wetness on carbonate building stones
Three moisture sensors were tested as a means for determining the surface wetness on carbonate building stones exposed to conditions that produce deposition of moisture. A relative-humidity probe, a gypsum-coated circuit grid, and a limestone-block resistor were tested as sensors for determining surface wetness. Sensors were tested under laboratory conditions of constant relative humidity and tempAuthorsR.B. See, M.M. Reddy, R. G. MartinInterpretation with a Donnan-based concept of the influence of simple salt concentration on the apparent binding of divalent ions to the polyelectrolytes polystyrenesulfonate and dextran sulfate
It has been shown that the apparent enhancement of divalent metal ion binding to polyions such as polystyrenesulfonate (PSS) and dextran sulfate (DS) by decreasing the ionic strength of these mixed counterion systems (M2+, M+, X-, polyion) can be anticipated with the Donnan-based model developed by one of us (J.A.M.). Ion-exchange distribution methods have been employed to measure the removal by tAuthorsJ.A. Marinsky, Robert F. Baldwin, M.M. ReddyProton and metal ion binding to natural organic polyelectrolytes-I. Studies with synthetic model compounds
A unified physico-chemical model, based on a modified Henderson-Hasselbalch equation, for the analysis of ion complexation reactions involving charged polymeric systems is presented and verified. In this model pH = pKa+p(??Ka) + log(??/1 - ??) where Ka is the intrinsic acid dissociation constant of the ionizable functional groups on the polymer, ??Ka is the deviation of the intrinsic constant dueAuthorsJ.A. Marinsky, M.M. ReddyProton and metal ion binding to natural organic polyelectrolytes-II. Preliminary investigation with a peat and a humic acid
We summarize here experimental studies of proton and metal ion binding to a peat and a humic acid. Data analysis is based on a unified physico-chemical model for reaction of simple ions with polyelectrolytes employing a modified Henderson-Hasselbalch equation. Peat exhibited an apparent intrinsic acid dissociation constant of 10-4.05, and an apparent intrinsic metal ion binding constant of: 400 foAuthorsJ.A. Marinsky, M.M. ReddyCrystal growth of calcite from calcium bicarbonate solutions at constant PCO2 and 25°C: a test of a calcite dissolution model
A highly reproducible seeded growth technique was used to study calcite crystallization from calcium bicarbonate solutions at 25°C and fixed carbon dioxide partial pressures between 0.03 and 0.3 atm. The results are not consistent with empirical crystallization models that have successfully described calcite growth at low PCO2 (< 10−3 atm). Good agreement was found between observed crystallizationAuthorsMichael M. Reddy, Niel Plummer, E. Busenberg - Software
Below are software products associated with this project.


