Non-lethal Detection of Skin Injuries in Juvenile Chinook Salmon Oncorhynchus tshawytscha by Fast Green FCF Dye Active
In fish, as in humans, an intact epidermis is critical to defense against entry of pathogens into the skin. Macroscopic examination of scale loss is the principal method of evaluating physical damage to juvenile salmonids out-migrating through hydroelectric dams in the Snake and Columbia Rivers, and in fish subjected to capture and handling procedures in locations such as hatcheries, fish bypass facilities at dams, or streams. However, this method cannot detect damage to the transparent epidermal tissue external to the scales, or in non-scaled areas such as the fins and head. It also cannot discern recent injuries from older injuries in which the epidermis has healed but scales have not yet regenerated. Improved non-lethal methods for evaluating the extent and timing of skin injuries could assist resource managers in identification of causes of injury, and in making modifications to bypass facilities or capture and handling methods to minimize injuries to fish.
Scientists at the USGS Western Fisheries Research Center (WFRC) developed a rapid non-lethal method for detecting recent skin and fin injuries in fish examined in the field. Although non-lethal application of fluorescein dye has been used to detect recent skin injuries in fish (similar to procedures for detection of eye injuries in humans), the fluorescein method requires use of an ultraviolet light in a darkened area, reducing its practicality for field use. The USGS procedure uses fast green FCF, an artificial food coloring that has also been used as a cell viability stain and requires no specialized equipment for observation of stained (injured) areas.
Fast green FCF staining of fish skin employs the principle of membrane permeability dye exclusion to rapidly distinguish healthy cells (where intact membranes exclude the dye) from dead or injured cells (which absorb the stain) in living fish. Unlike human skin, all layers of the epidermis of the skin of most fish species are living tissue and will exclude fast green FCF when the epidermis is intact. Immediately after injury, the dead tissue will stain, but following closure of the wound by migrating epidermal cells, staining is no longer observed. Depending on water temperature, migrating epidermal cells can close small surface wounds (less than 5 mm) within 3 to 24 hours, but regeneration of scales may not be evident for a minimum of 8 to 10 days after injury.
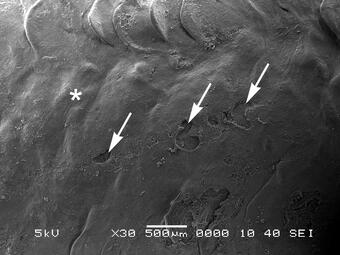
The WFRC scientists found that a 1-minute exposure of juvenile Chinook salmon to fast green FCF diluted to a concentration of 0.1% in water was sufficient to stain recent skin and fin injuries so that they could be easily observed (Figures 1 and 3). Examination of skin tissue sections by scanning electron microscopy confirmed that the dye stained only the open wounds that lacked epidermis (Figure 2), and that wounded areas no longer stained once the epidermal layer was re-established, even if no scale regeneration was yet visible (Figure 4). Further studies of juvenile Chinook salmon held in freshwater and seawater after dye exposure showed no significant differences in survival or skin healing in fish that had been exposed to fast green FCF or no dye after wounding.
The experiments in our laboratory indicated that fast green FCF staining can be used as a rapid non-lethal method to identify sites of recent epidermal injury in juvenile Chinook salmon and can augment the descaling analyses presently used in the field.



Below are publications associated with this project.
Evaluation of fast green FCF dye for non-lethal detection of integumental injuries in juvenile Chinook salmon Oncorhynchus tshawytscha
Below are partners associated with this project.
Scanning electron microscopy training and consultation were provided by Carla Stehr and Brian Bill of NOAA/Northwest Fisheries Science Center.
In fish, as in humans, an intact epidermis is critical to defense against entry of pathogens into the skin. Macroscopic examination of scale loss is the principal method of evaluating physical damage to juvenile salmonids out-migrating through hydroelectric dams in the Snake and Columbia Rivers, and in fish subjected to capture and handling procedures in locations such as hatcheries, fish bypass facilities at dams, or streams. However, this method cannot detect damage to the transparent epidermal tissue external to the scales, or in non-scaled areas such as the fins and head. It also cannot discern recent injuries from older injuries in which the epidermis has healed but scales have not yet regenerated. Improved non-lethal methods for evaluating the extent and timing of skin injuries could assist resource managers in identification of causes of injury, and in making modifications to bypass facilities or capture and handling methods to minimize injuries to fish.
Scientists at the USGS Western Fisheries Research Center (WFRC) developed a rapid non-lethal method for detecting recent skin and fin injuries in fish examined in the field. Although non-lethal application of fluorescein dye has been used to detect recent skin injuries in fish (similar to procedures for detection of eye injuries in humans), the fluorescein method requires use of an ultraviolet light in a darkened area, reducing its practicality for field use. The USGS procedure uses fast green FCF, an artificial food coloring that has also been used as a cell viability stain and requires no specialized equipment for observation of stained (injured) areas.
Fast green FCF staining of fish skin employs the principle of membrane permeability dye exclusion to rapidly distinguish healthy cells (where intact membranes exclude the dye) from dead or injured cells (which absorb the stain) in living fish. Unlike human skin, all layers of the epidermis of the skin of most fish species are living tissue and will exclude fast green FCF when the epidermis is intact. Immediately after injury, the dead tissue will stain, but following closure of the wound by migrating epidermal cells, staining is no longer observed. Depending on water temperature, migrating epidermal cells can close small surface wounds (less than 5 mm) within 3 to 24 hours, but regeneration of scales may not be evident for a minimum of 8 to 10 days after injury.
The WFRC scientists found that a 1-minute exposure of juvenile Chinook salmon to fast green FCF diluted to a concentration of 0.1% in water was sufficient to stain recent skin and fin injuries so that they could be easily observed (Figures 1 and 3). Examination of skin tissue sections by scanning electron microscopy confirmed that the dye stained only the open wounds that lacked epidermis (Figure 2), and that wounded areas no longer stained once the epidermal layer was re-established, even if no scale regeneration was yet visible (Figure 4). Further studies of juvenile Chinook salmon held in freshwater and seawater after dye exposure showed no significant differences in survival or skin healing in fish that had been exposed to fast green FCF or no dye after wounding.
The experiments in our laboratory indicated that fast green FCF staining can be used as a rapid non-lethal method to identify sites of recent epidermal injury in juvenile Chinook salmon and can augment the descaling analyses presently used in the field.




Below are publications associated with this project.
Evaluation of fast green FCF dye for non-lethal detection of integumental injuries in juvenile Chinook salmon Oncorhynchus tshawytscha
Below are partners associated with this project.
Scanning electron microscopy training and consultation were provided by Carla Stehr and Brian Bill of NOAA/Northwest Fisheries Science Center.

