Intestinal obstruction with Porrocaecum sp. ascarids in a Snowy Owl (Bubo scandiacus)
Snowy owl visitor to CCGS Louis S. St-Laurent in the Arctic Ocean
History: An adult, female 1130-g Snowy Owl (Bubo scandiacus) was found dead in a field in Wisconsin, USA and submitted for diagnostic necropsy.
Gross Findings: On external examination, the owl was emaciated and had numerous feather lice. The right eye was swollen and red-brown material in the anterior chamber partly obscured the iris. There was a 5 mm diameter ulcer in the skin over the keel, and an open fracture of the right hallux (first toe).
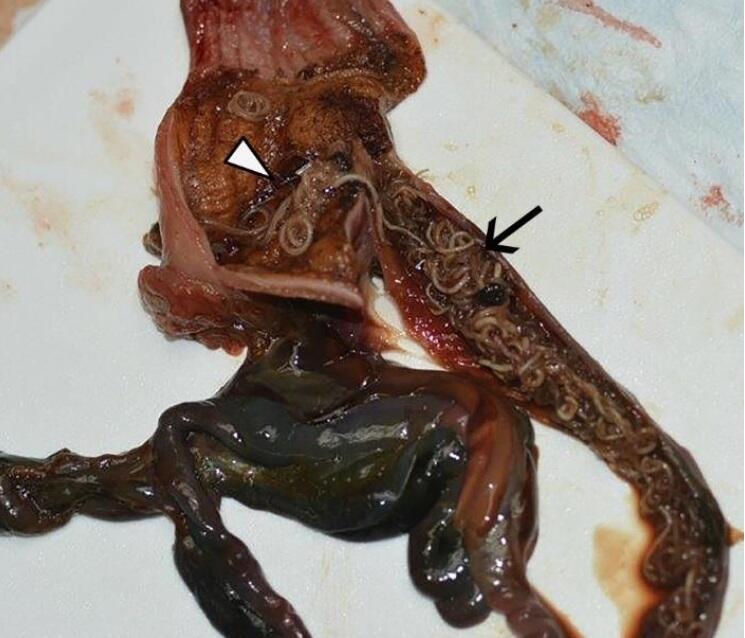
On internal examination, there was no subcutaneous, pericardial, or epicardial fat. The proventriculus and duodenum contained numerous (>100), long, pale tan nematodes that distended and occluded the proximal duodenum (Fig. 1). Mucosa of the duodenum was dark red.
Sources/Usage: Photo: photograph by Anna Panike
Figure 1. Photograph from a Snowy Owl (Bubo scandiacus) found dead in Wisconsin, USA. Numerous nematodes are present in the ventriculus (white arrowhead) and duodenum (black arrow).
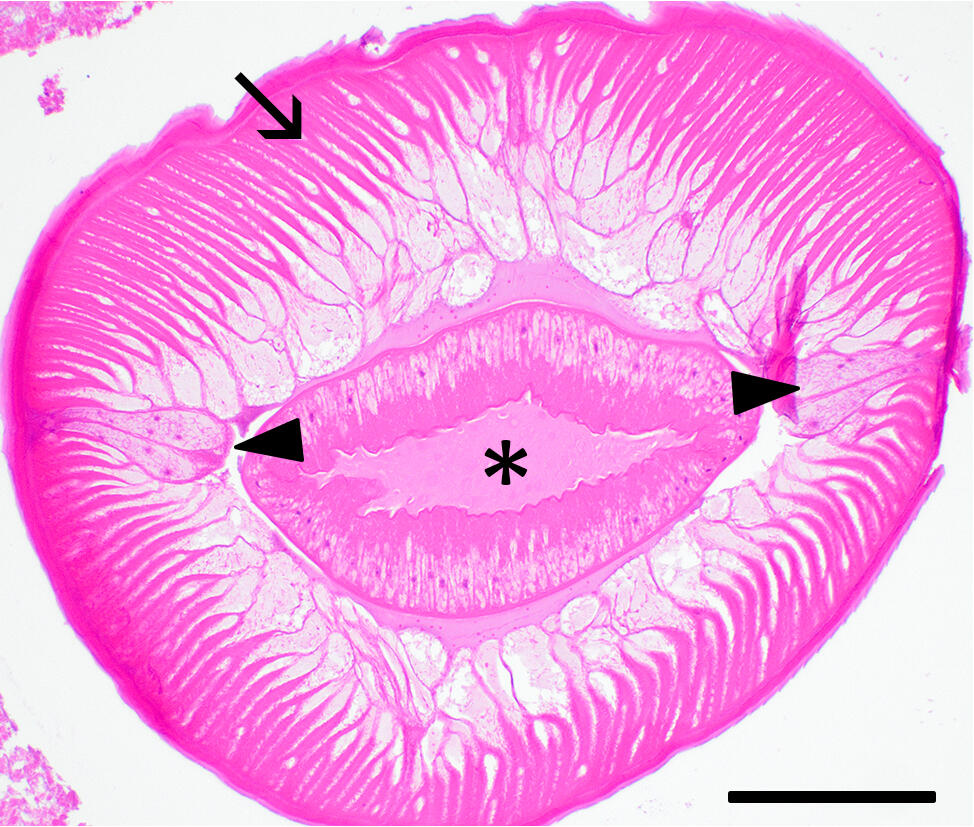
Histopathological Findings: Large nematode cross sections are present within the lumen of the jejunum, mixed with nematode eggs and necrotic debris. Nematodes have a finely ridged cuticle with small lateral internal ridges, prominent lateral chords, polymyarian coelomyarian musculature, and an intestine lined by tall columnar epithelium (Fig. 2).
Figure 2. Cross section of nematode showing tall polymyarian-coelomyarian musculature (arrow), prominent lateral chords (arrowheads) and intestine (asterisk). Bar = 200 µm.
Etiologic Diagnosis: Intestinal nematodiasis with starvation and obstruction.
Disease: Intestinal porrocaecosis
Etiology: Porrocaecum is a genus of ascarid nematodes found in birds and occasionally in mammals. Species taxonomy is in flux; species are difficult to distinguish morphologically, and molecular data is sparse.
Distribution: Global.
Seasonality: No seasonal correlation.
Host range: Porrocaecum spp. have been described from the digestive tracts of birds across every continent including Antarctica (Sanjota and others, 2013). The taxonomy of species within the genus is in flux, and while around 40 species are described, the validity of many of those is in question. Only one species has been completely sequenced and seven more have incomplete genetic information recorded. (Gu and others, 2023).
Transmission: In the cases where a life cycle is known, Porrocaecum spp. develop within an earthworm intermediate host that is infected by consuming eggs in the soil. Eggs may then encyst in the tissues of small mammals that eat infected earthworms. Birds of prey are then infected when they consume the small mammals (Al-jehni, 2008).
Clinical signs: With low or moderate levels of infection there may be no clinical signs, with higher levels, weight loss, ruffled feathers, or starvation may be seen.
Pathology: In most cases of infection there is little to no pathogenic effect. With concurrent stress and high intensity infections, intestinal inflammation from worms embedded in the mucosa, peritonitis (if worms perforate intestinal walls), and/or starvation can be seen. (Al-jehni, 2008).
Diagnosis: Worms may be identified in the feces of infected birds or seen in the intestines at necropsy. Speciation requires sequencing, however, only one species has been completely sequenced for reference.
Public health concerns: Unknown, but likely very low risk. Porrocaecum spp. infections have not been described in humans, but related genera (e.g. Contraceacum) have been found in mammals as aberrant hosts. Mammals, including humans, are not infected directly by consuming eggs and humans do not usually eat earthworms, which are the intermediate host.
Wildlife population impacts: None. In some instances 40% of a population is infected with no measurable effect (Presswell and Bennett, 2024).
Management: None.
References:
Al-jehni, M., 2008, Parasitic diseases of wild bords: Ames, Iowa, Wiley-Blackwell.
Gu, X.-H., Guo, N., Chen, H.-X., Sitko, J., Li, L.-W., Guo, B.-Q., and Li, L., 2023, Mitogenomic phylogenies suggest the resurrection of the subfamily Porrocaecinae and provide insights into the systematics of the superfamily Ascaridoidea (Nematoda: Ascaridomorpha), with the description of a new species of Porrocaecum: Parasites & Vectors, v. 16, no. 1, p. 275.
Presswell, B., and Bennett, J., 2024, Gastrointestinal helminths of the Australasian harrier (Circus approximans Peale, 1848) in New Zealand, and description of a new species of nematode, Procyrnea fraseri n. sp. (Habronematidae): Journal of helminthology, v. 98, p. e6.
Sanjota, U.M.S., Das, N., Channa, M.A., and Khan, M.M., 2013, New host record of the genus Porrocaecum Rallet & Henry, 1912 (Nematoda: Anisakidae) frm the bird Egretta garzetta in Hyderbad, Sindh, Pakistan. : Pakistan Journal of Nematology, v. 31, no. 2, p. 183-186.