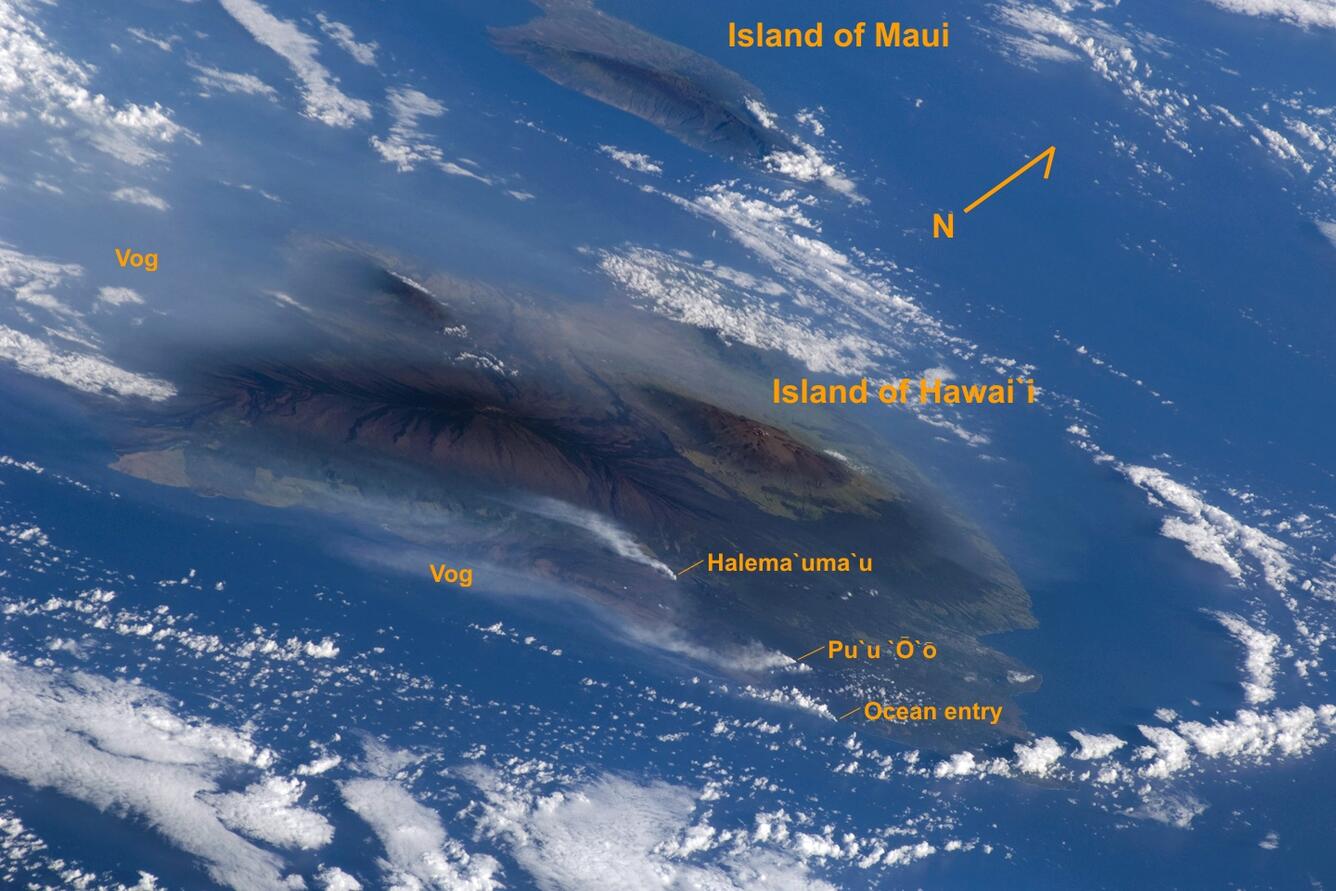
Volcanic gas emissions are composed mainly of water vapor (H2O), carbon dioxide (CO2), and sulfur dioxide (SO2) gas, with trace amounts of several other gaseous compounds, including hydrogen sulfide (H2S), hydrogen fluoride (HF), and carbon monoxide (CO). The chief gas hazard in Hawai‘i results from SO2 gas.

Vog (volcanic air pollution) affects broad areas downwind of erupting vents
Vog is a hazy mixture of SO2 gas and aerosols, which are primarily composed of sulfuric acid droplets and other sulfate (SO4) compounds. Aerosols are created when SO2 and other volcanic gases combine in the atmosphere and interact chemically with oxygen, moisture, dust, and sunlight over periods of minutes to days. The particles, or particulate matter (PM), in vog, are characterized as PM2.5 because the particles are less than 2.5 micrometers in diameter—less than 1/20th the width of a human hair. PM2.5 are considered to be 'fine' particles, which can travel deeper into human lungs than larger particles. Vog particles grow by absorbing water vapor and other gases, so they can increase in size in a moist environment such as the human upper respiratory tract (nose, mouth, and throat).
The exact composition of vog depends on how much time the volcanic plume has had to react in the atmosphere. In areas such as the Kona coast on the leeward side of Hawai‘i, far from the erupting vents at Kīlauea, aerosols are the main component of vog. Closer to the volcano, vog contains both aerosols and unreacted SO2 gas.
Learn more: Hawaii Interagency Vog Information Dashboard
People with pre-existing medical conditions are the primary group at risk of experiencing health effects from exposure to vog, but healthy people may also experience symptoms.
Agricultural crops and other plants are subject to injury by exposure to the pollutants that emerge from Kīlauea, especially when they are present in high concentrations. Farmers and gardeners in the path of the pollutants (SO2 and acid rain) have reported significant damage to plants caused by winds blowing SO2 gas and acid particles from Pu‘u ‘Ō‘ō and Halema‘uma‘u to downwind areas.
Many homes on the Island of Hawai‘i rely on rooftop rainwater-catchment systems for their household water supply. In 1988, the drinking water from 40 percent of home catchment systems in the Kona Districts of the island were found to be contaminated with lead leached from roofing and plumbing materials, such as nails, paint, solder, and metal flashings, by acid rain.
Read more about the impact of vog on health, agriculture, and water-catchment systems on the Hawaii Interagency Vog Information Dashboard.
Style of eruption controls SO2 gas emissions
Top: From 1983–86, spectacular but short-lived lava fountains occurred about once every three weeks at the Pu‘u ‘Ō‘ō vent on the East Rift Zone. The wind cleared the air of volcanic gases and particles between the eruptive episodes.
Middle: From 1986-2018, the character of the eruption changed to a nearly continuous effusion of lava and gas. Although the vent location has shifted many times since 1986, gas release has been essentially constant. With no opportunity for the air to clear, vog became a persistent presence on the Island of Hawai‘i. The Kupaianaha shield lava pond is pictured on April, 8, 1987.
Bottom: From 2008-2018, a continuously erupting vent in Halema‘uma‘u Crater at the summit of Kīlauea increased the overall amount of SO2 released from Kīlauea and the amount of vog impacting the State of Hawaii.
Volcano Watch articles about vog, SO2, and CO2 emissions
- Dual nature of vog results in air quality variety show
- Reduced sulfur dioxide emissions from Kīlauea's east rift have some fans cheering
- In every volcanic paradise, a little vog must fall
- Murky crystal ball suggests potential for voggy future at Kīlauea
- A volatile view of CO2 from atop Kīlauea's magma chamber
Lava entering ocean creates localized air pollution (laze)
When molten lava flows into the ocean, it reacts vigorously with sea water to create a different type of gas plume that results in hazy and noxious conditions downwind of an ocean entry. Referred to as a "laze" plume (for a blending of the words 'lava' and 'haze'), it forms through a series of chemical reactions as hot lava boils seawater to dryness. The plume is an irritating mixture of hydrochloric acid gas (HCl), steam, and tiny volcanic glass particles. This hot, corrosive gas mixture caused two deaths immediately adjacent to the coastal entry point in 2000, when seawater washed across recent and active lava flows. Visitors should avoid this plume at the entry point and downwind, as even the wispy edges of it can cause skin and eye irritation and breathing difficulties. Acid rain from the plume has a pH between 1.5 and 3.5, and has the corrosive properties of dilute battery acid.
The ocean-entry plume is blown in various directions by wind. Downslope air flow from nighttime through early morning typically blows the laze plume off shore and out to sea. Between mid-morning and late afternoon, however, trade wind conditions blows the plume along the coast and inland, resulting in locally poor air quality.
Volcano Watch articles about laze hazards
Volcanic gas emissions are composed mainly of water vapor (H2O), carbon dioxide (CO2), and sulfur dioxide (SO2) gas, with trace amounts of several other gaseous compounds, including hydrogen sulfide (H2S), hydrogen fluoride (HF), and carbon monoxide (CO). The chief gas hazard in Hawai‘i results from SO2 gas.

Vog (volcanic air pollution) affects broad areas downwind of erupting vents
Vog is a hazy mixture of SO2 gas and aerosols, which are primarily composed of sulfuric acid droplets and other sulfate (SO4) compounds. Aerosols are created when SO2 and other volcanic gases combine in the atmosphere and interact chemically with oxygen, moisture, dust, and sunlight over periods of minutes to days. The particles, or particulate matter (PM), in vog, are characterized as PM2.5 because the particles are less than 2.5 micrometers in diameter—less than 1/20th the width of a human hair. PM2.5 are considered to be 'fine' particles, which can travel deeper into human lungs than larger particles. Vog particles grow by absorbing water vapor and other gases, so they can increase in size in a moist environment such as the human upper respiratory tract (nose, mouth, and throat).
The exact composition of vog depends on how much time the volcanic plume has had to react in the atmosphere. In areas such as the Kona coast on the leeward side of Hawai‘i, far from the erupting vents at Kīlauea, aerosols are the main component of vog. Closer to the volcano, vog contains both aerosols and unreacted SO2 gas.
Learn more: Hawaii Interagency Vog Information Dashboard
People with pre-existing medical conditions are the primary group at risk of experiencing health effects from exposure to vog, but healthy people may also experience symptoms.
Agricultural crops and other plants are subject to injury by exposure to the pollutants that emerge from Kīlauea, especially when they are present in high concentrations. Farmers and gardeners in the path of the pollutants (SO2 and acid rain) have reported significant damage to plants caused by winds blowing SO2 gas and acid particles from Pu‘u ‘Ō‘ō and Halema‘uma‘u to downwind areas.
Many homes on the Island of Hawai‘i rely on rooftop rainwater-catchment systems for their household water supply. In 1988, the drinking water from 40 percent of home catchment systems in the Kona Districts of the island were found to be contaminated with lead leached from roofing and plumbing materials, such as nails, paint, solder, and metal flashings, by acid rain.
Read more about the impact of vog on health, agriculture, and water-catchment systems on the Hawaii Interagency Vog Information Dashboard.
Style of eruption controls SO2 gas emissions
Top: From 1983–86, spectacular but short-lived lava fountains occurred about once every three weeks at the Pu‘u ‘Ō‘ō vent on the East Rift Zone. The wind cleared the air of volcanic gases and particles between the eruptive episodes.
Middle: From 1986-2018, the character of the eruption changed to a nearly continuous effusion of lava and gas. Although the vent location has shifted many times since 1986, gas release has been essentially constant. With no opportunity for the air to clear, vog became a persistent presence on the Island of Hawai‘i. The Kupaianaha shield lava pond is pictured on April, 8, 1987.
Bottom: From 2008-2018, a continuously erupting vent in Halema‘uma‘u Crater at the summit of Kīlauea increased the overall amount of SO2 released from Kīlauea and the amount of vog impacting the State of Hawaii.
Volcano Watch articles about vog, SO2, and CO2 emissions
- Dual nature of vog results in air quality variety show
- Reduced sulfur dioxide emissions from Kīlauea's east rift have some fans cheering
- In every volcanic paradise, a little vog must fall
- Murky crystal ball suggests potential for voggy future at Kīlauea
- A volatile view of CO2 from atop Kīlauea's magma chamber
Lava entering ocean creates localized air pollution (laze)
When molten lava flows into the ocean, it reacts vigorously with sea water to create a different type of gas plume that results in hazy and noxious conditions downwind of an ocean entry. Referred to as a "laze" plume (for a blending of the words 'lava' and 'haze'), it forms through a series of chemical reactions as hot lava boils seawater to dryness. The plume is an irritating mixture of hydrochloric acid gas (HCl), steam, and tiny volcanic glass particles. This hot, corrosive gas mixture caused two deaths immediately adjacent to the coastal entry point in 2000, when seawater washed across recent and active lava flows. Visitors should avoid this plume at the entry point and downwind, as even the wispy edges of it can cause skin and eye irritation and breathing difficulties. Acid rain from the plume has a pH between 1.5 and 3.5, and has the corrosive properties of dilute battery acid.
The ocean-entry plume is blown in various directions by wind. Downslope air flow from nighttime through early morning typically blows the laze plume off shore and out to sea. Between mid-morning and late afternoon, however, trade wind conditions blows the plume along the coast and inland, resulting in locally poor air quality.