Distemper Keratitis and Pneumonia and Baylisascaris in a Raccoon (Procyon lotor)
History: In January 2025 a young raccoon was observed acting lethargic and sick near a K-12 school in Wisconsin, USA. The raccoon was euthanized and submitted to the U.S. Geological Survey National Wildlife Health Center (NWHC) for diagnosis.
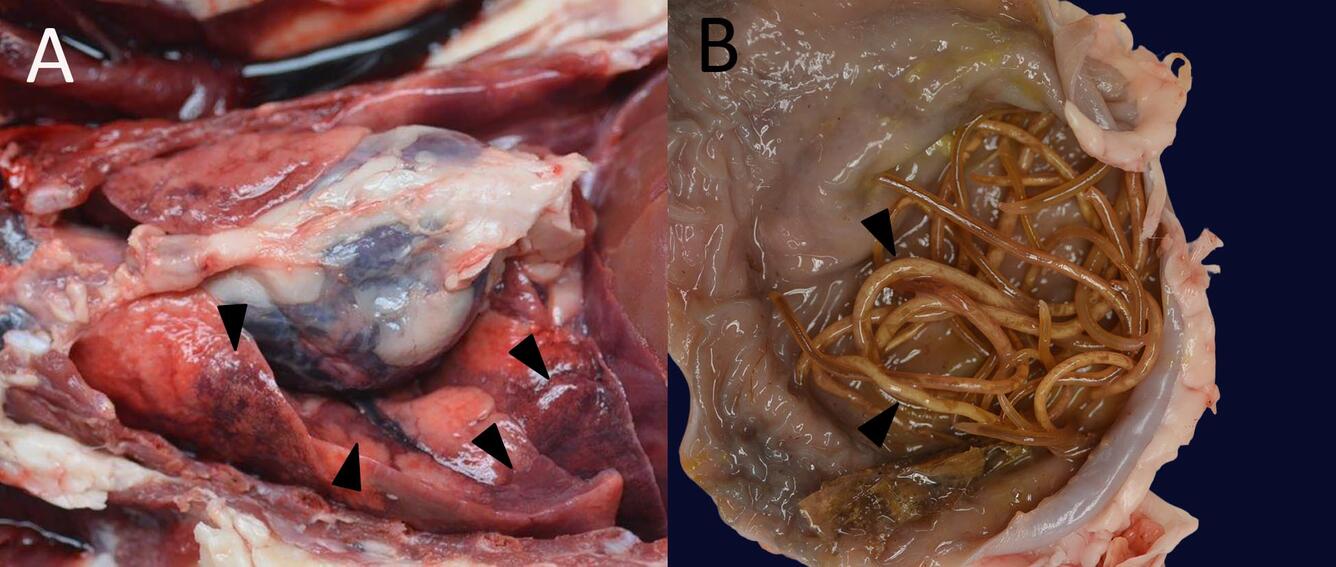
Gross Findings: At necropsy, the raccoon was a young female in excellent nutritional condition. The right cornea was cloudy, and the left cornea was opaque, pale blue, and collapsed. The lungs were heavy and wet, and the left caudal lung lobe was moderately overinflated, heavy, and mottled dark red (Fig. 1A). The stomach contained one piece of slightly woody vegetation and numerous, 10-20 cm long, pale yellow nematodes (Fig. 1B) with several similar nematodes found within the small intestine.
Sources/Usage: Public Domain. Photos by Anna Panike.
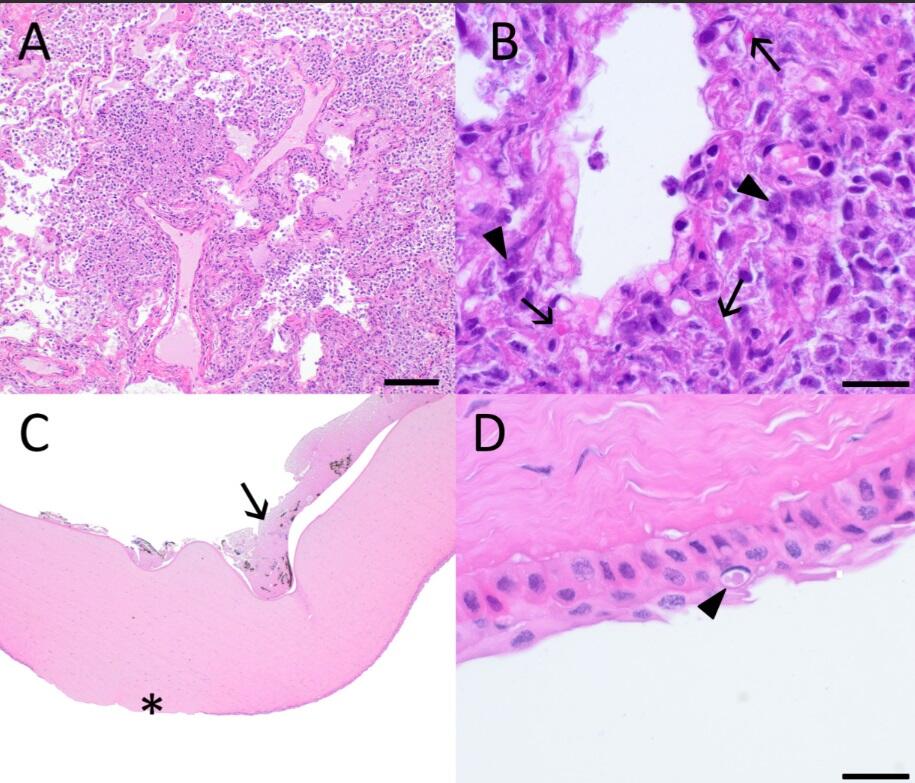
Histopathological Findings: Within the lung there was marked to severe, multifocal coalescing pyogranulomatous bronchointerstitial pneumonia that sometimes obliterates alveolar architecture (Fig. 2A). Occasional syncytial cells are present, and bright eosinophilic, amorphous, intracytoplasmic and intranuclear, variably sized viral inclusions are found within bronchial and alveolar epithelial cells and macrophages (Fig. 2B). Multifocal areas of necrosis are present, which contain clusters of coccobacteria and large numbers of degenerate and nondegenerate neutrophils.
Within the left eye, the corneal epithelium was ulcerated and eroded, with moderate edema of underlying stroma (Fig. 2C). Rare epithelial cells contained eosinophilic, variably sized, intracytoplasmic or intranuclear inclusions (Fig. 2D). Corneal endothelium was necrotic and sloughing, and fibrin and eosinophilic fluid containing few neutrophils, macrophages and both free and phagocytosed pigment were present in the anterior chamber (Fig 2C).
Histology:
Morphologic Diagnoses:
- Lung. Acute bronchointerstitial pneumonia, pyogranulomatous, multifocal coalescing, marked, with intracytoplasmic and intranuclear viral inclusion bodies.
- Eye. Ulcerative keratitis and anterior uveitis, locally extensive, moderate, with rare intracytoplasmic and intranuclear viral inclusions.
- Stomach and small intestine. Baylisascaris nematodiasis.
Ancillary tests and results: PCR using lung tissue for canine distemper virus (CDV) was positive. A representative nematode from the stomach is morphologically consistent with Baylisascaris procyonis.
A direct fluorescent antibody assay for the detection of rabies virus inclusion bodies in brain tissue conducted by the Wisconsin Hygiene Communicable Disease Division, and Avian influenza matrix rRT-PCR on nasal and rectal swabs were negative.
Disease: Canine distemper, B. procyonis
Etiology: Canine distemper virus is a morbillivirus within the family Paramyxoviridae, is closely related to both rinderpest (eliminated in 2011) and the virus that causes human measles. Similar but distinct viruses also infect small ruminants, and marine mammals (Morens and others, 2011).
Baylisascaris procyonis, the raccoon roundworm, is a large ascarid nematode. Prevalence in raccoon populations is high, up to 80% in some reports, but morbidity in raccoons is minimal (Gu and others, 2024).
Distribution: Canine distemper has a worldwide distribution.
The distribution of Baylisascaris procyonis mirrors that of raccoons throughout North America and is an emerging concern in other continents where raccoons have been introduced (Xie and others, 2014; Lombardo and others, 2022).
Seasonality: CDV and raccoon roundworm are found year-round, but increased prevalence of both distemper and B. procyonis can be seen in early spring through summer when kits are born and raised, due to the high susceptibility of young animals.
Host range: CDV has a wide host range that includes domestic dogs and wild canids and has been reported in most carnivore families including large cats, skunks, weasels and otters, bears, hyenas, seals and sea lions, civets, and red pandas (Ailurus fulgens). It has also been reported in wild collared peccaries (Dicotyles tajacu), in marmots and macaques in captivity, and in experimentally-induced infections in pigs and mice (Martinez-Gutierrez and Ruiz-Saenz, 2016).
Raccoons are the definitive host for Baylisascaris, but dogs can also serve as patent hosts and many mammals, including humans may become aberrant hosts.
Transmission: Transmission of canine distemper virus is through aerosol droplets or body fluids. Transmission of Baylisascaris is fecal-oral or by ingestion of paratenic hosts (e.g. mice).
Clinical signs: Raccoons with distemper may be noted wandering during the day and may demonstrate circling, ataxia, seizures, ocular and nasal discharge, difficulty breathing, and diarrhea. Hyperkeratosis of the foot pads and nasal planum can be seen in animals that survive the acute period.
Baylisascaris in raccoons is usually asymptomatic but may cause diarrhea and weight loss in young animals with high loads.
Pathology: CDV enters through the upper respiratory tract, proliferates in regional lymph nodes, and then spreads to multiple organs with a tropism for lymphoid, epithelial, hematopoietic and neuroectodermal cells. Generalized lymphoid depletion is common. Interstitial pneumonia from viral infection of pulmonary epithelial cells can be complicated by suppurative pneumonia due to secondary bacterial infections resulting from lymphoid depletion and immune suppression. Catarrhal enteritis, keratoconjunctivitis and uveitis can also be seen, as well as nonsuppurative meningoencephalitis, gliosis, and demyelination in the brain. Amorphous, 1 to 5 µm, eosinophilic, cytoplasmic and/or intranuclear viral inclusion bodies may be found in multiple organs, and syncytial cells are often present. Virus may persist in surviving animals in the eye, brain and in dendritic cells, and some animals can become persistent shedders. In surviving dogs this is implicated in the development of demyelinating leukoencephalitis “old dog encephalitis” years later (Beineke and others, 2009; Origgi and others, 2012), but is not known whether a similar sequela occurs in raccoons.
Diagnosis: Diagnosis in live animals can be via PCR or serology, but rabies is an important differential, so wild mammals with neurologic signs should be euthanized and tested at an accredited laboratory. Diagnosis of canine distemper at necropsy is based on compatible gross lesions and characteristic histology, followed by immunohistochemistry demonstrating viral antigen within lesions or positive PCR.
Baylisascaris can be diagnosed by fecal floatation or examination of adult worms found at necropsy. PCR and sequencing are necessary for definitive identification, but a strong presumptive ID can be made based on morphology and host species.
Public health concerns: CDV is not known to infect humans, but vaccination can be used to protect pet dogs and ferrets.
Baylisascaris procyonis can infect a wide range of aberrant hosts, including humans, where migrating larvae may invade various tissues, including brain and eye, with devastating and potentially fatal results. Young children who have not yet learned hand washing hygiene have the highest risk (Sorvillo and others, 2002). Dogs can also be patent hosts, so protecting dogs that live in areas where raccoons are common with a monthly parasiticide that covers roundworms also serves to reduce the risk to humans.
Wildlife population impacts: CDV is responsible for periodic mortality events in many carnivore species and can have significant impact on imperiled or endangered species such as the black footed ferret (Williams and others, 1988), and losses due to distemper infection contributed to the extinction of the Thylacine (Paddle, 2012).
The range of Baylisascaris has expanded with the introduction of raccoons to areas in Europe and Asia. Its wide paratenic host range presents the potential for unknown impacts on naive species (Lombardo and others, 2022).
Management: Carcasses and sick animals should be removed from the area during an outbreak. Vaccination is effective at preventing disease, and standard disinfectants will decontaminate surfaces in wildlife or zoo facilities. Strict isolation with attention to air handling is important in managing cases in zoo or wildlife hospitals, as CDV is highly transmissible via aerosol.
References:
Beineke, A., Puff, C., Seehusen, F., and Baumgärtner, W. 2009 Pathogenesis and immunopathology of systemic and nervous canine distemper. Vet Immunol immunop. 127(1): 1-18.
Gu, X.H., Chen, H.X., Hu, J.J., and Li, L. 2024. Morphology and ASAP analysis of the important zoonotic nematode parasite Baylisascaris procyonis (Stefahski and Zarnowski, 1951), with molecular phylogenetic relationships of Baylisascaris species (Nematoda: Ascaridida): Parasitology: 151(2), 200-212.
Lombardo, A., Brocherel, G., Donnini, C., Fichi, G., Mariacher, A., Diaconu, E.L., Carfora, V., Battisti, A., Cappai, N., Mattioli, L., and De Liberato, C. 2022. First report of the zoonotic nematode Baylisascaris procyonis in non-native raccoons (Procyon lotor) from Italy. Parasite Vector: 15( 1) p. 24.
Martinez-Gutierrez, M., and Ruiz-Saenz, J. 2016. Diversity of susceptible hosts in canine distemper virus infection: a systematic review and data synthesis: BMC Vet Res. 12: 78.
Morens, D.M., Holmes, E.C., Davis, A.S., and Taubenberger, J.K. 2011. Global rinderpest eradication: lessons learned and why humans should celebrate too. J Infect Dis.204(4): 502-505.
Origgi, F.C., Plattet, P., Sattler, U., Robert, N., Casaubon, J., Mavrot, F., Pewsner, M., Wu, N., Giovannini, S., Oevermann, A., Stoffel, M.H., Gaschen, V., Segner, H., and Ryser-Degiorgis, M.-P. 2012. Emergence of Canine Distemper Virus Strains With Modified Molecular Signature and Enhanced Neuronal Tropism Leading to High Mortality in Wild Carnivores: Vet Path. 49(6): 913-929.
Paddle, R. 2012. The thylacine's last straw: epidemic disease in a recent mammalian extinction. Aust Zool. 36(1): 75-92.
Sorvillo, F., Ash, L.R., Berlin, O.G., and Morse, S.A. 2002. Baylisascaris procyonis: an emerging helminthic zoonosis: Emerg Infect Dis. 8(4): 355-359.
Williams, E.S., Thorne, E.T., Appel, M.J., and Belitsky, D.W. 1988. Canine distemper in black-footed ferrets (Mustela nigripes) from Wyoming: J Wildl Dis. 24(3): 385-398.
Xie, Y., Zhou, X., Li, M., Liu, T., Gu, X., Wang, T., Lai, W., Peng, X., and Yang, G. 2014. Zoonotic Baylisascaris procyonis roundworms in raccoons, China. Emerg Infect Dis. 20(12) 2170-2172.