Emaciation with serous atrophy of fat in a Northern Sea Otter (Enhydra lutris)
History: A subadult, male Northern Sea Otter (Enhydra lutris) was found recently dead on a beach in Kodiak Island, Alaska and submitted for diagnostic necropsy.
Gross Findings: On external examination, eyes were sunken, and bones were prominent on palpation. On internal examination there was no subcutaneous, visceral, perirenal or pericardial fat, and observed (cervical, abdominal) musculature was atrophic. The stomach contained a small piece of plastic film, and intestines were empty of ingesta.
Histopathological Findings: Fat cells in adipose tissues within mesenteries, surrounding lymph nodes, and at the renal hila are shrunken, and many contain vacuolated eosinophilic cytoplasm with residual lipid and enlarged, central to eccentric, rather than marginalized nuclei. Germinal centers are indistinct and reduced in size and number within the adjacent lymph node, and lymphocytes are depleted.
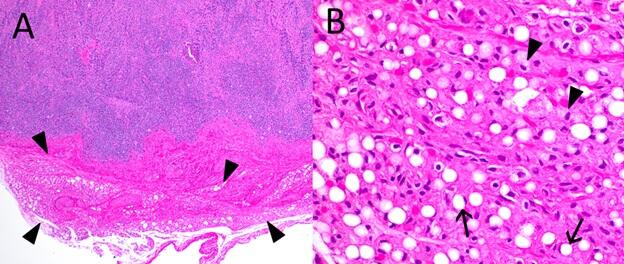
Figure 1. Photomicrographs from a Northern Sea Otter (Enhydra lutris) found dead on a beach in Alaska, USA. (A) Lymph node. Germinal centers are depleted. Adjacent mesenteric adipose tissue markedly atrophic (arrowheads). (B) High magnification of mesenteric adipose tissue. Adipocytes are shrunken, with reduced lipid storage vacuoles (arrows) or have frothy eosinophilic cytoplasm and central to eccentric nuclei (arrowheads).
Morphologic diagnosis:
Subcutaneous, pericardial, perirenal and mesenteric diffuse, severe, fat atrophy, with generalized, moderate skeletal muscle atrophy (emaciation).
Mesenteries – Diffuse, severe serous atrophy of fat.
Lymph node – Diffuse, marked lymphoid depletion.
Etiology: Starvation.
Seasonality: Starvation and adipose tissue atrophy can be seen in any animal that is not able to meet energetic demands due to environmental conditions or debilitation from prolonged illness or chronic injury. In this animal, no other findings compatible with injury, chronic infection, or toxicity were found during the postmortem examination. Young, newly weaned animals of any species are at increased risk as the transition period from maternal care to self-sufficiency is challenging, so emaciated young animals may be seen during the time that they are typically weaned or shortly after. Sea otters have a very high basal metabolic rate, and the energetic demands of pup rearing for the mother are nearly double that of pre-pregnancy levels by the average weaning age (Thometz and others, 2014).
Clinical signs: Weakness and lethargy; emaciated body condition may be difficult to appreciate under thick fur or feathers. Animals that are not meeting caloric requirements may eat non-food items or wander into unusual areas in search of food.
Pathology: As stored lipids are mobilized in the face of nutritional deficit, adipose cells shrink and change from being filled by a single, large lipid vacuole that displaces the nucleus to eosinophilic to pale grey cytoplasm with small vacuoles of residual lipid. These can resemble brown fat or even signet ring carcinoma on histology (Gilbert and others, 2021). Grossly, areas where adipose tissue was formerly more abundant appear gelatinous. Fat around the heart and kidneys is the last to be metabolized in starvation, and absence of fatty tissue in these locations is also evidence of a chronic, severe nutritional deficit. Germinal centers in lymphoid organs are often depleted and malnourished animals are immune compromised (Chandra, 1996). Muscle wasting occurs as fat stores are consumed. Death may result from infection due to immune compromise, or from metabolic acidosis and electrolyte imbalance leading to cardiac arrest.
Diagnosis: Diagnosis is based on overall body condition, absence of pericardial, perirenal and mesenteric fat, and characteristic histology.
Public health concerns: Similar changes are seen in humans with starvation or cachexia due to illness (e.g. cancer). Hungry animals may enter more populated areas in search of food, increasing the risk of conflicts with humans.
Wildlife population impacts: Starvation is a major cause of otter mortality in early life. In a survey of Northern Sea Otters in Washington and Oregon, emaciation without other lesions was the second most common cause of death for immature and subadult sea otters (tied with trauma from all causes), behind Sarcocystis encephalitis as the most common cause (White and others, 2018). Otters do not develop fat stores for insulation or as a buffer for periods of low caloric intake like other marine mammals (e.g. seals) and depend on daily intake. Weanling otters have a high metabolic rate and may not have developed the skills for successful foraging. If prey is sparse, the mother may also abandon a pup or wean earlier for her own survival (Cortez and others, 2016). Poor body condition also contributes to a peak in adult female deaths during the period around weaning (Thometz and others, 2014).
Management: Prey availability in an area is a major determinant for the feasibility and success of reintroduction programs (USFWS, 2022). Abandoned pups may be raised in captivity and possibly released back to the wild if found alive, but supplemental feeding of wild otters is not done because it can increase human directed aggression and decrease foraging behavior.
References:
Chandra, R.K., 1996, Nutrition, immunity and infection: from basic knowledge of dietary manipulation of immune responses to practical application of ameliorating suffering and improving survival: Proceedings of the National Academy of Sciences, v. 93, no. 25, p. 14304-14307.
Cortez, M., Wolt, R., Gelwick, F., Osterrieder, S.K., and Davis, R.W., 2016, Development of an altricial mammal at sea: I. Activity budgets of female sea otters and their pups in Simpson Bay, Alaska: Journal of Experimental Marine Biology and Ecology, v. 481, p. 71-80.
Gilbert, J.D., Langlois, N.E.I., Ruszkiewicz, A., Moffat, D., and Cash, K., 2021, Signet-ring cell appearance of atrophic fat cells: Forensic Science, Medicine and Pathology, v. 17, no. 1, p. 141-143.
Thometz, N.M., Tinker, M.T., Staedler, M.M., Mayer, K.A., and Williams, T.M., 2014, Energetic demands of immature sea otters from birth to weaning: implications for maternal costs, reproductive behavior and population-level trends: Journal of Expeimental Biology, v. 217, no. Pt 12, p. 2053-2061.
USFWS, 2022, Feasabliity assessment: Sea otter reintroduction to the Pacific coast, Report to Congress prepared by the U.S. Fish and Wildlife Service, Region 9; and Regions 10, Sacramento, California.
White, C.L., Lankau, E.W., Lynch, D., Knowles, S., Schuler, K.L., Dubey, J.P., Shearn-Bochsler, V.I., Isidoro-Ayza, M., and Thomas, N.J., 2018, Mortality trends in northern sea otters (Enhydra lutris kenyoni) collected from the coasts of Washington and Oregon, USA (2002–15): Journal of Wildlife Diseases, v. 54, no. 2, p. 238-247.