Comprehensive Sturgeon Research Project Blog - 2016
The USGS Comprehensive Sturgeon Research Project is a multi-year, interdisciplinary research study to determine factors leading to spawning and survival of the endangered pallid sturgeon and the closely related shovelnose sturgeon.
Click here to return to the 2021 blog
Remote Technology Monitors Sturgeon Spawning
By Aaron DeLonay, Kimberly Chojnacki and Chad Vishy
August 3, 2016
Scientists from the U.S. Geological Survey and their partners have been successful in using telemetry to track adult sturgeon to their spawning locations on the Missouri and Yellowstone Rivers for several years (see previous blog entry A Spawning Recorded in the Yellowstone River). Despite locating spawning locations at a relatively fine resolution (10’s to 100’s of m), we continue to struggle to observe spawning and egg deposition in the swift turbid water. The definition of spawning habitat suitability and functionality is dependent upon adequately identifying quality spawning habitat (see previous blog entry Spawning patches on the Yellowstone River.) Scientists at the Columbia Environmental Research Center (CERC) are conducting studies to adapt new methods and technology to study spawning behavior and more precisely locate and characterize areas selected by pallid sturgeon for egg deposition. These studies will hopefully allow scientists to assess what characteristics of functional spawning habitats attract adults and result in the survival of developing embryos. This would help to both understand why sturgeon are having difficulty reproducing successfully and provide guidance for enhancing or creating functional spawning habitats.
One way in which scientists at CERC do this is to work with the closely related shovelnose sturgeon. We have implanted test shovelnose sturgeon with miniature acoustic transmitters and release them into specially constructed experimental mesocosms at CERC (see previous blog entry We Just Wanna See What Happens Down There!). Circulators in the mesocosm provide a continuous current that simulates conditions in the Missouri River. Scientists can manipulate the substrate in the mesocosms to provide patches of either gravel or cobble where sturgeon may spawn. We map fish locations using an array of 8 to 16, precisely arranged hydrophones connected by cables to a telemetry receiver. The hydrophones detect the acoustic pulses of the transmitters in the sturgeon. The telemetry receiver records each transmitted pulse and uses the signal detected by the array of hydrophones to calculate the precise position of each sturgeon in the pond, as often as once per second. In some trials an ARIS multibeam acoustic camera has been placed in the center of the pond to allow researchers to record sturgeon behavior.


The spawning trials begin when female and male sturgeon are primed with hormone injections and released into the pond to let nature take its course. Scientists can then evaluate the performance of the miniature transmitters by watching the tracks of individually tagged fish as the males and females move, come together, and finally spawn by releasing eggs and milt. Comparing the ARIS sonar imagery with the precise tracks from the telemetry system allows scientists to identify movement patterns that indicate spawning and egg deposition. If the transmitters perform well in the mesocosm and spawning behavior can be identified through patterns of movement, then the transmitters could be used in the wild fish in the river to determine the precise timing and location of spawning, and the habitat characteristics where egg deposition occurs. Scientists can then better study if these spawning patches are functional, and whether the quality of the patches used by sturgeon is limiting embryo development and survival.


River of sand
July 26, 2016
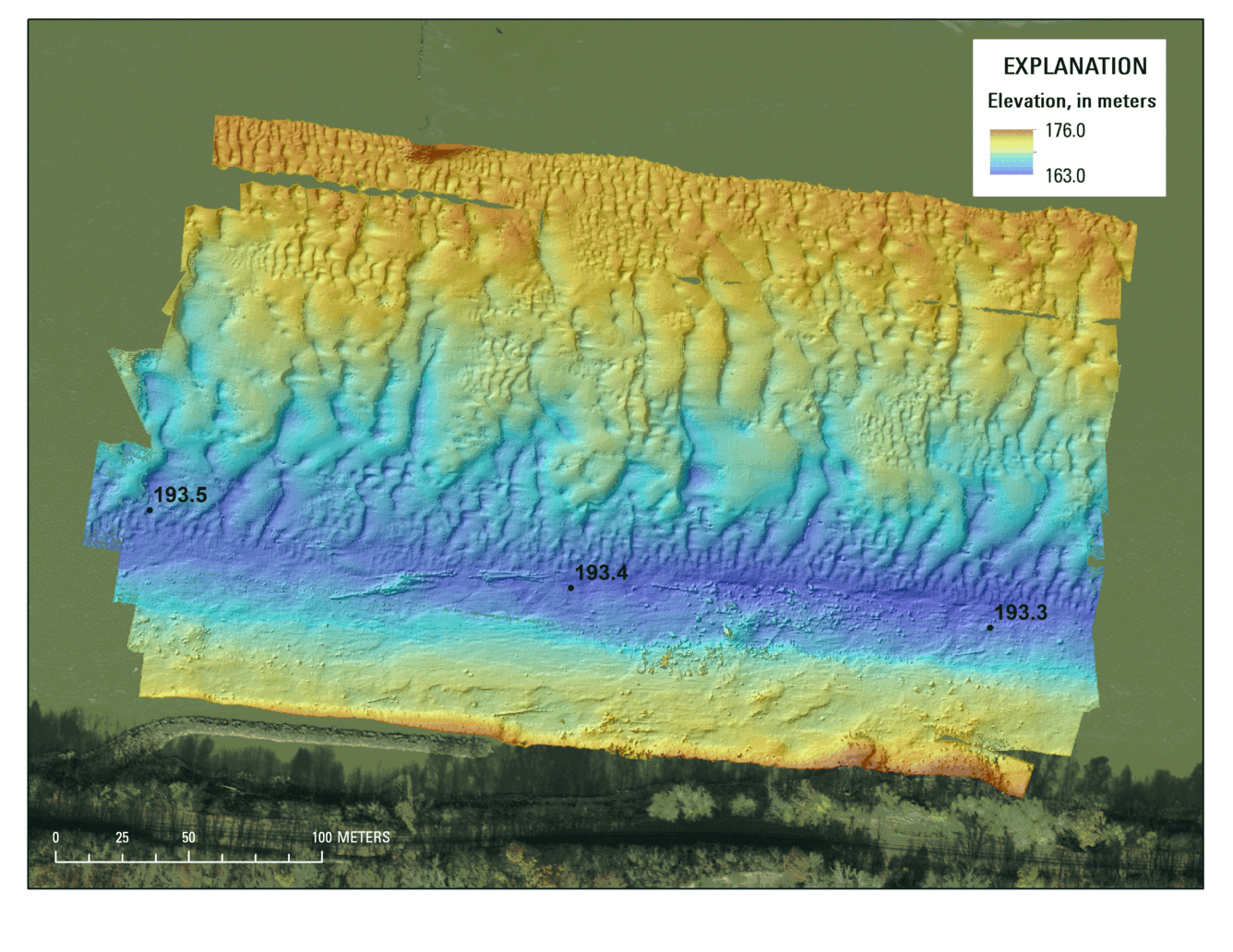
Pallid sturgeon are benthic fish, which means they live near the bottom of the river. We know from hydroacoustic mapping that the bottom of the Missouri River is mostly made of sand, and that in much of the channel the sand dunes are moving rather quickly; at times up to 3 meters per hour! Pallid sturgeon navigate through and use sand dunes and the flow fields around dunes when they migrate upstream to spawn. It is also highly likely that that sand dunes affect feeding patterns through macroinvertebrate drift and that river substrate type and disturbance regime is related to macroinvertebrate food production in the river. We’ve been using a multibeam echosounder and precise GPS positioning to survey the river, monitor change, and measure rates of sand dune movement. These measurements help us to understand how environments on the bottom of the river that pallid sturgeon use change with flow, and if the places on the river where sturgeon spawn (revetment and rocky bedrock outcrops like you can see in Figure 1) are stable or instead subject to episodic erosion and deposition.
Transitioning from One Life Stage to the Next
July 20, 2016
The release of nearly 700,000 pallid sturgeon free embryos on June 27 (see previous blog entry 700,000 Baby Fish) marked the start of the most spatially extensive drift and dispersal study ever conducted in the Missouri River basin. Following release of the newly hatched pallid sturgeon, crews started to sample for the drifting free embryos on June 27, first at a site only a few miles downstream. The sampling regime moved progressively downstream as the free embryos drifted, and sampling continued round-the-clock (see previous blog entry Night Sampling for Pallid Sturgeon Free Embryos). Two sites (one near Culbertson, MT at river mile 1620 and the other at Williston, ND at river mile 1550) were sampled for multiple continuous 24-hr cycles in an attempt to characterize the entire distribution of drifting free embryos arriving, passing, and trailing slowly through these sampling sites. On July 8, nearly 12 days after the free embryos were released 210 miles upstream, sampling for free embryos that might still be drifting came to an end as the U. S. Fish and Wildlife Service team of Ryan Wilson and Sam Hultberg cranked the net winches for the last time, flushed the paired larval nets in the flowing river near Williston, and searched the detritus for any potential pallid sturgeon free embryos remaining in the drift.
Does that last pull of larval nets on July 8 mark completion of the free embryo study? Not at all! Rather, it completes one aspect of the study – sampling for free embryos as they disperse downstream with the current. Next, a second and equally important aspect of the study starts. Growth and development of surviving free embryos (see previous blog entry A Change is Gonna Come) eventually leads to settlement, the developmental process by which free embryos transition from drifting to associating with (settling on) river bed habitats as larvae. Following exhaustion of their yolk-supply feeding reserves, the settled larvae start to feed on invertebrates in the river. With termination of drift sampling, other crews initiated the second important aspect of the study focused on capturing larval pallid sturgeon that may have survived and settled on the river bed.
Crews on July 7 and July 8 sampled multiple sites with a benthic beam trawl, spanning a 24-mile reach extending from river mile 1560 (upstream from Williston) down to about river mile 1536 (downstream from Williston in the flowing headwaters of Lake Sakakawea). The trawling of multiple habitats yielded much of what was expected – young channel catfish, sicklefin chubs, and a smattering of other species. But, beam trawling also produced five small sturgeon, ranging in length from 31-41 mm. Two of the small sturgeon were sampled at river mile 1536. Because pallid sturgeon and shovelnose sturgeon at this small size are visually nearly identical, genetic testing is required to differentiate the two species. Further genetic testing is then required to demonstrate that a captured pallid sturgeon larvae is one of the 700,000 released upstream.

Beam trawling through a large reach of the Missouri River and the headwaters of Lake Sakakawea will continue for several weeks in an attempt to capture young pallid sturgeon originally released as free embryos, and to determine where settlement and survival may have happened. All small (e.g., < 130 mm) sturgeon collected will be subjected to genetic testing. Along with the field work, laboratory work continues as personnel process drift samples, and sort and count pallid sturgeon free embryos and beads collected during the drift-sampling framework.

It cannot be overstated – dedication to this project has been outstanding. All individuals – ranging from hatchery personnel for their superb fish culture practices to the fish samplers for their long day and night shifts – should be commended for their great work in support of this highly relevant scientific information.
A Change is Gonna Come
By Kimberly Chojnacki, and Aaron DeLonay
July 14, 2016
With all the discussion of free embryos this summer, some readers may be left wondering what, exactly, is a ‘free embryo’. A free embryo is a developing fish no longer within a protective chorion (egg envelope), from the time of hatch to the initiation of active feeding. This life stage is marked by rapid development, growth, and change. Newly hatched pallid sturgeon free embryos are generally 7-9 millimeters long with a large yolk-sac to fuel the rapid development and growth of the tiny fish. The free embryos hatch without a well-developed mouth, eyes, gills, fins, or the fleshy, whiskerlike barbels near the mouth of sturgeon (figure 1). Without fins, a newly hatched pallid sturgeon free embryo has limited ability to control its movement in the river current for several days after hatch. Water temperature is considered to be the most important environmental factor influencing the development of this early life stage.

At approximately 20 °C and 2 days after hatch, eyes of the free embryos have become pigmented, and pectoral fins buds, and barbel buds are visible (figure 2).

By approximately the fifth day after hatch, the free embryos are developing gill filaments, a dorsal fin, and the barbels continue to grow (figure 3).

At approximately 20 °C and 10 days after hatch, the pallid sturgeon free embryos look much more like miniature versions of their parents with a broad, flat rostrum (snout or nose), fleshy barbels bordering the protrusible mouth, and large pectoral fins (figure 4). The free embryos are now approximately 17-19 millimeters long and are ready to begin active feeding and transition to the larval stage.

Lake Sakakawea Delta and Headwaters
July 13, 2016

On July 5, the USGS acoustic Doppler current profiler crews worked from just south of Williston, North Dakota, downstream to the headwaters of Lake Sakakawea. The objective of this part of the study was to characterize velocities and habitat complexity in the part of the system that is thought to be fatal to pallid sturgeon free embryos. A seminal paper by Dr. Chris Guy and colleagues established in 2015 that low dissolved oxygen in the headwaters of Fort Peck Lake was the most likely cause of mortality for pallid sturgeon free embryos in that part of the Upper Missouri River system (Guy and others, 2015). This conclusion has been broadly accepted to hold true for Lake Sakakawea as well. Research into the potential anoxic zone in Lake Sakakawea is continuing through Montana State University; our data are intended to provide some additional physical context to help understand the role of anoxia in pallid sturgeon ecology.


The effect of the lake on river processes is quite evident. Upstream near Fort Peck Dam, the river banks are high, and often bare and eroding, indicating that the channel is incising. Moving downstream through the delta and headwaters, the bank height decreases, sediment deposition becomes apparent, and habitats become more complex. A big difference is the presence of gently sloping banks and different ages of floodplain willows and cottonwoods, indicating relatively frequent access of water to the floodplain. Floodplain connectivity is generally associated with high ecosystem productivity.
Reference:
Guy, C.S., Treanor, H.B., Kappenman, K.M., Scholl, E.A., Ilgen, J.E., and Webb, M.A.H., 2015, Broadening the regulated-river management paradigm: A case study of the forgotten dead zone hindering pallid sturgeon recovery: Fisheries, v. 40, no. 1, p. 6–14. 10.1080/03632415.2014.987236.
Steady Flows and Collaborative Science
July 6, 2016
One of the key components to the Upper Missouri River Pallid Sturgeon Drift Experiment (see previous blog entry Pallid Sturgeon Free Embryo Drift Experiment Starts) was steady release of flow from Fort Peck Dam. Usually this time of year, releases cycle daily to follow electrical demand. Those so-called peaking cycles would have made the drift experiment more difficult to carry out and much more difficult to evaluate. The effect of the steady flow releases can be seen in a graph of water stage at the US Geological Survey streamflow gaging station just downstream from Fort Peck dam (figure 1).

In the weeks before the experiment, stages varied by about a foot whereas during the experiment flow releases resulted in fluctuations of less than 0.15 foot. The collaborators on the Drift Experiment are very grateful for the assistance of the U.S. Army Corps of Engineers Missouri River Basin Water Management Division and the Western Area Power Administration in keeping flow releases steady during the duration of the experiment.
Velocity Measurements in Support of the Drift Study
By Robb Jacobson, Ph.D. and Susannah Erwin, Ph.D.
July 3, 2016
One of the key components of the Upper Missouri River Pallid Sturgeon Drift Study is the collection of detailed water-velocity information. Water velocities are used to calibrate the advection-dispersion model (see previous blog entry Models and baby fish – digits, dye, particles, and biological reality) and to develop understanding of how channel form may affect transport and retention of pallid sturgeon free embryos.
We use boat-mounted acoustic Doppler current profilers (ADCP) to measure 3-dimensional velocity fields in transects oriented across the channel (figures 1 and 2). Positioning and navigation are provided through differentially corrected global positioning systems (GPS) that allow us to collect data in precise, pre-determined locations. The velocity data quantify the average velocity as well as the cross-sectional variation in velocity. Cross-sectional variation indicates where a river reach is likely to transport or retain dispersing free embryos (the life stage from hatch through first feeding).


The original sample design was to measure velocities at intervals along 221 miles of the Missouri River while the pallid sturgeon free embryos dispersed downstream, but river conditions have shown that we couldn’t traverse long sections of the river as rapidly as planned (see previous blog entry Boating the Upper Missouri River is not for the faint of heart). We revised our sampling design on the fly to emphasize detailed data collection in the reach upstream from Wolf Point, Montana, to support analysis of the dye trace component (see previous blog entry Missouri River Dye Trace Experiment to Support Understanding of Free Embryo Drift). From Wolf Point Montana to Lake Sakakawea we intend to provide a statistical subsample of the river (figure 3). Time is a constraint because we need to complete data collection while the US Army Corps of Engineers and Western Area Power Administration maintain steady releases from Fort Peck Dam.

The statistical subsample is intended to quantify the variation in channel condition and velocity distributions that exist along the Upper Missouri River. Complex and wide reaches are expected to have more variable velocity distributions (figure 4) and more retention potential compared to straight, narrow reaches (figure 5).


Night sampling for pallid sturgeon free embryos (The Fish Don’t Sleep and Neither Do We)
By Robb Jacobson, Ph.D. and Aaron DeLonay
July 2, 2016
The sample design for the Upper Missouri River Pallid Sturgeon Drift Experiment depends on nearly around-the-clock sampling for free-embryos, the life stage from hatch to the initiation of feeding. It can be a grueling process under the best of conditions, but for some of the crews there is the added complication of sampling at night. The challenges of wind, rain, and swarming insects are intensified in the chill of a dark Montana night.
The night sampling effort starts at about sunset – 8 pm in this part of Montana in the summer – and continues 12 hours until crews are relieved the next morning (figure 1). The crews transit to the pre-selected sampling sites before it gets too dark to avoid grounding on sandbars (see previous blog entry Boating the Upper Missouri River is not for the faint of heart).

Sampling requires winching a pair of heavily weighted, very fine-mesh nets (in figure 2) to the bottom of the Missouri River for a 10-minute collection, then winching it back up, emptying the contents of the net into a shallow sorting pan, and then sending the nets down again as quickly as possible. The nets sample just above the bottom of the river and collect a large amount of detritus and organic matter. The crew quickly picks through the contents of the nets for free-embryo sturgeon and experimental beads. The free embryos are counted and retained for genetic analysis while the number and color of beads are recorded. The up and down cycle of the nets repeats all night.

It’s an intensive and strenuous process, and a bit eerie working from the light of headlamps while anchored in the middle of the river (figure 3). In this type of sampling there are many net hauls that have zero free embryos or beads, so the rewards are not always immediate. The zero catches are important, however, because they indicate that the experimental release has not yet reached a site, or that all of the free embryos have passed. When the first free embryos and beads show up in the samples (figure 4), the shouts of success carry in the night from one headlamp to the next, and across the river to the companion boat anchored in the darkness—then the night is not so dark, nor as cold, as it was before the crews found what they were looking for.


Models and baby fish – digits, dye, particles, and biological reality
By Robb Jacobson, Ph.D. and Ed Bulliner
July 1, 2016
The Upper Missouri River Pallid Sturgeon Drift Study is ultimately about providing sound scientific information for smart decisions. The decisions relate to how to manage this large river to help recover the endangered pallid sturgeon, or at least to avoid doing additional harm. The Missouri River Recovery Management Plan (http://moriverrecovery.usace.army.mil/mrrp/f?p=136:70) has been developing new sources of science information to address how this can be done.
A prominent hypothesis for the lack of pallid sturgeon population growth in the Upper Missouri River is that there is insufficient drift distance downstream from Fort Peck dam. This idea holds that there is not enough river to allow for development of swimming and foraging capability for the drifting sturgeon free embryos (the period from hatch until the initiation of feeding) before they are swept into Lake Sakakawea where they may succumb to various causes of mortality, especially the threat of very low dissolved oxygen in the lake.
Dr. Craig Fischenich and colleagues in the US Army Corps of Engineers have developed a computational model – an advection/dispersion model – that can be used to address how river-management decisions change available and required drift distance. It’s a state-of-the-art model, but like any model it has assumptions and estimates. One of the main motivations for the Drift Study is to test the assumptions and find ways to improve the model. The multi-objective structure of the study is meant to determine how well drifting sturgeon free embryos conform to the model assumptions.
Our dye-trace sub-experiment (see previous blog entry Missouri River Dye Trace Experiment to Support Understanding of Free Embryo Drift) is meant to evaluate key components of the advection/dispersion model, in particular how well it predicts transport of a purely passive substance. Early download of fluorometer data(figure 1) from sites within 13 miles of the release site indicate that the dye is moving somewhat faster and is less spread out along the river compared to initial model estimates. Ultimately, passive transport will be compared to free embryo transport to develop biological reality for the model.

An important part of the model is the longitudinal dispersion coefficient that measures how particles spread out as they travel down the river. By varying that coefficient we can calibrate the model to more exactly replicate actual conditions. The comparisons between model results in red and dye trace data in blue, before (figure 2) and after calibration (figure 3), show the importance of the dye data. Final calibration of the model will require assessment of a lot more data, but initial calibrations have proven useful in determining where and when fish biologists should sample for pallid sturgeon free embryos during the study.


Boating the Upper Missouri River is not for the faint of heart
June 30, 2016
The Upper Missouri River is mostly left to its own devices, allowed to migrate as it pleases. As a result, the river provides complex habitat that may lead to retention and growth of larval pallid sturgeon. The complexity is also vexing for boat drivers because it means that opportunities to get stuck on sandbars are around every bend.

The boat of choice has an aluminum hull with a jet prop. On plane, these boats generally draft less than a couple of inches. Problems crop up when the water is shallower than that or the driver picks the wrong route in complex channels. The sandbars have a tendency to grab hard and hold fast. Getting a stranded boat off of a sandbar can require brains and brawn.

Knowing the river and being able to read the water are key. Using GPS and digital aerial photography, boat crews have even more ways to read the water, but we’ve found that images only two years old can be inaccurate in places where the river is especially active.
700,000 Baby Fish
June 28, 2016
On Monday afternoon, June 27, the collaborating scientists of the Missouri River Pallid Sturgeon Free Embryo Drift Study delivered over 700,000 one-day post hatch (dph) pallid sturgeon to the Upper Missouri River. The drift experiment has been over a year in the making and has been completely dependent on the ability to produce a statistically large number of newly hatched pallid sturgeon. Through the concerted effort of Rob Holm and staff at the U.S. Fish and Wildlife Service, Garrison Dam National Fish Hatchery, 7 reproductive females and 5 males were synchronized to bring off the mass hatch. The day-old sturgeon were then trucked from Garrison to the Upper Missouri River, about 2 miles downstream of the Milk River Confluence (see previous blog entry Pallid Sturgeon Free Embryo Drift Experiment Starts) by U.S. Fish and Wildlife personnel (figure 1).

Once the tiny fish were acclimated to the ambient water temperature, they were released in the river to track their downstream dispersal (figure 2). A small armada of sampling boats has been mobilized to capture these fish as they drift down the river over the next week. Ultimately, the results of this experiment are intended to show how the fish spread out in the river, how far they disperse, and whether they are retained in route.

The downstream sampling effort has been spearheaded by Pat Braaten, U.S. Geological Survey, and Dave Fuller and Tyler Haddix of Montana Fish Wildlife and Parks. They are joined by a navy of fisheries biologists from U.S. Geological Survey, Montana Fish Wildlife and Parks, U.S. Fish and Wildlife Service, and Nebraska Game and Parks Commission. The sampling will continue – night and day – for as many as 7 to 9 days until it appears that all fish have been transported to Lake Sakakawea or have been retained in supportive habitats. The samplers are especially grateful to the U.S. Army Corps of Engineers and the Western Area Power Administration for arranging to keep flows steady during the experiment.
Missouri River Dye Trace Experiment to Support Understanding of Free Embryo Drift
June 27, 2016
Missouri River scientists started the first phase of the pallid sturgeon free embryo drift study by employing a standard method to understand how hydraulic processes vary the rate of downstream transport of dissolved constituents and particles. On June 26 they released 50 liters of rhodamine-WT dye (figure 1) into the Missouri River about 10 miles downstream of Fort Peck Dam, Montana (about 1.75 miles downstream of the Milk River confluence). The downstream dispersion of dye is being recorded through a network of fluorometers distributed through about 84 miles of the river. Rhodamine-WT is a harmless but very visible dye (figure 2) that is frequently used to study time of travel in river systems, usually to assess contaminant transport rates.


The experiment is being helped a great deal by collaboration with US Army Corps of Engineers and Western Area Power Administration, who have arranged for near-steady flows for the duration of the experiment.
The initial effect of putting the dye in the river is dramatic, as nearly the entire river turned bright red (figure 3). With continuous dilution, the bright red faded to burnt umber after drifting about 6 miles. After 20 miles it was no longer visually identifiable, although the instruments have been able to detect the dye to concentrations as low as 1 parts per billion.

Information from the dye trace is being used in near-real-time to refine the sampling strategy for the companion free embryo-drift experiment taking place in the afternoon of June 27. More about the collaborative free embryo drift experiment in subsequent posts.
Pallid Sturgeon Free Embryo Drift Experiment Starts
By Robb Jacobson, Ph.D. and Casey Hickcox
June 26, 2016
Missouri River scientists started a large-scale collaborative study on June 24 to improve understanding of the fate of pallid sturgeon free embryos (from hatch through first feeding) in the Upper Missouri River (figure 1). The study is being led by scientists from the U.S. Geological Survey with strong collaboration with Montana Fish Wildlife and Parks, U.S. Fish and Wildlife, U.S. Army Corps of Engineers, Bureau of Reclamation, and Western Area Power Administration. The ambitious study will use pallid sturgeon free embryos produced at Garrison National Fish Hatchery as tracers to test dispersal models and better understand transport dynamics.

Results of previous studies indicate that pallid sturgeon free embryos need to drift long distances before they can safely settle out of the water column and begin feeding. Better understanding of the dynamics in which the free embryos move downstream, and how long they need to safely settle out and begin to feed, may lead to important recovery options for this endangered fish. Crews will follow the free embryos downstream twenty-four hours a day for over a week, using ichthyoplankton nets to sample the drifting population.
The free embryo drift experiment is being complemented by assessment of river hydraulics. Scientists from the USGS Columbia Environmental Research Center are collecting an extensive collection of velocity data using acoustic Doppler current profilers, in concert with a rhodamine dye trace. The dye trace is intended to quantify the dispersion processes that spread free embryos out along the river through retention. In addition, collaborators with Montana Fish Wildlife and Parks are using tiny plastic beads as a tracer; the beads are designed to mimic free embryo settling behavior. If the beads adequately mimic drifting free embryos, further studies could eliminate the need to use live sturgeon; saving time, money and resources. Other boat crews will be measuring the hydraulics of the Missouri River at a very high resolution to gain a better understanding of how these tiny fish move through the differing habitat conditions.
Sturgeon biology and plumbing
By Robert Jacobson, Ph.D., Aaron DeLonay, Kimberly Chojnacki, and Casey Hickcox
May 23, 2016
This spring, Columbia Environmental Research Center (CERC) scientists are conducting laboratory experiments to better understand how temperature affects early development of sturgeon species. The emphasis is on quantifying development rates from fertilization of eggs to first-feeding larvae. Previous experiments at CERC have explored relations between temperature and development but they had always been limited in the size and scope by laboratory facilities. To more fully explore early development of sturgeon species, CERC needed a new, updated, and larger version of our fish hatching system.
The new apparatus, designed by CERC biologist James Candrl and built by biologist (and master plumber) Dave Combs, offers scientists 24 temperature-controlled hatching and holding environments for the experiments, allowing for 6 different temperature treatments with 4 replicates. The partially-recirculating systems can use either local well water or a reconstituted water made in large volumes at CERC to simulate any water quality where sturgeon may hatch and develop. The system circulates the water through ultraviolet sterilizers to remove disease organisms and control fungus. The sterilized water is routed through an electronically-controlled heating and chilling units that maintain the desired temperatures. The heated and chilled water is then pumped up and allowed to trickle down through large degassing columns that remove excess dissolved gasses that can accumulate and saturate the water when the temperature is manipulated, potentially harming young fish. The degassed water is then routed to the hatching jars where the fertilized eggs are gently rolled by the moving water during incubation. The hatching jars are constructed so hatched free embryos automatically overflow into holding tanks where they reside until they are ready to be transferred into rearing tanks and initiate feeding.

The new system provides great flexibility for evaluating development rates as a function of water temperature, and for comparing different sturgeon species and different genetic parentage within species. Experiments planned for this spring include pallid sturgeon (Scaphirhynchus albus) and the closely related, the shovelnose sturgeon (Scaphirhynchus platorynchus).

Experiments on Early Sturgeon Development
By Robert Jacobson, Ph.D., Aaron DeLonay, Kimberly Chojnacki, and Casey Hickcox
May 20, 2016
Pallid sturgeon (Scaphirhynchus albus), like most fish, are cold-blooded. This means that their metabolism is regulated by the temperature of the water in which they live. As water warms, developmental rates of sturgeon free embryos generally increase, up to a point where water becomes too warm and then they start to die. If the water is too cold, then the free embryos cannot reach critical developmental milestones and they will also die. In some parts of the Missouri River there is potential to influence water temperature — and possibly development and survival of early pallid sturgeon — by altering the way water is released from dams. Management of dam releases to influence early sturgeon development presumes a rigorous, quantitative understanding of the relation between water temperature, sturgeon development, and the transition of sturgeon from one life stage (free embryos) to the next (benthically-oriented, feeding larvae).
Experiments at CERC this spring are intended to increase understanding the effect of temperature on free embryo development . A new hatching system allows for precise temperature control and multiple treatments to evaluate hatch and free-embryo development. In the first implementation this spring, four female pallid sturgeon were induced to spawn and their eggs were collected and manually fertilized using milt from three males.

Figure 1 CERC Biologist, James Candrl, checks to determine if a female pallid sturgeon is releasing eggs .
Each family of eggs was separated into replicate environments to have their development monitored at treatments with water temperatures ranging 14 – 26o C. The data collected relate the rate at which free-embryo sturgeon reach critical developmental milestones with cumulative temperature experienced by the fish over time. The experiments are designed to create a model to predict developmental transitions based upon cumulative temperature units and to explore how developmental trajectories vary through the entire range of river temperatures that pallid sturgeon spawn.

New Pallid Sturgeon Reports
January 29, 2016
Our blog has been quiet lately because we have been concentrating on writing and reporting. It has been said that if scientific information isn’t published, it’s as if it doesn’t exist. On rivers like the Missouri, where contentious management issues create a high demand for quality science, there is a special need to publish high-quality, peer-reviewed information in a timely manner. Our project scientists have been catching up during the last several months, striving to complete a backlog of progress reports on pallid sturgeon ecology. We have recently completed publication of 5 of these reports, including annual reports for 2012 and 2013, and 3 reports for the Effects Analysis; our 2014 annual report is being finalized and should be published in winter 2016. The 2012 annual report was a special effort to synthesize progress from studies 2005 through 2012.

The USGS’ investment in a high-quality peer review process delays publications somewhat, but results in scientific information products that can be considered credible, authoritative, and unbiased. The USGS production process assures that well-edited reports are published into the public domain and are freely available to stakeholders and the public.
These reports are available at the indicated pubs.usgs.gov websites:
DeLonay, A.J., Jacobson, R.B., Chojnacki, K.A., Braaten, P.J., Buhl, K.J., Eder, B.L., Elliott, C.M., Erwin, S.O., Fuller, D.B., Haddix, T.M., Ladd, H.L.A., Mestl, G.E., Papoulias, D.M., Rhoten, J.C., Wesolek, C.J., and Wildhaber, M.L., 2016, Ecological requirements for pallid sturgeon reproduction and recruitment in the Missouri River: Annual report 2013: U.S. Geological Survey Open-File Report 2015–1197, 99 p. https://pubs.er.usgs.gov/publication/ofr20151197
DeLonay, A.J., Chojnacki, K.A., Jacobson, R.B., Albers, J.L., Braaten, P.J., Bulliner, E.A., Elliott, C.M., Erwin, S.O., Fuller, D.B., Haas, J.D., Ladd, H.L.A., Mestl, G.E., Papoulias, D.M., and Wildhaber, M.L., 2016, Ecological requirements for pallid sturgeon reproduction and recruitment in the Missouri River: A synthesis of science, 2005-2012: U.S. Geological Survey, Scientific Investigations Report 2015-5145, 224 p. https://pubs.er.usgs.gov/publication/sir20155145
Jacobson, R.B., Parsley, M.J., Annis, M.L., Colvin, M.E., Welker, T.L., and James, D.A., 2015, Development of conceptual ecological models linking management of the Missouri River to pallid sturgeon population dynamics: U.S. Geological Survey, Open-File Report 2015-1038, 47 p. https://pubs.er.usgs.gov/publication/ofr20151038
Jacobson, R.B., Parsley, M.J., Annis, M.L., Colvin, M.E., Welker, T.L., and James, D.A., 2016, Development of working hypotheses linking management of the Missouri River to population dynamics of Scaphirhynchus albus (pallid sturgeon): U.S. Geological Survey, Open-file Report 2015-1236, 33 p. https://pubs.er.usgs.gov/publication/ofr20151236
Jacobson, R.B., Annis, M.L., Parsley, M.J., James, D.A., Colvin, M.E., and Welker, T.L., 2016, Scientific information to support the Missouri River pallid sturgeon effects analysis: U.S. Geological Survey, Open-file Report 2015-1226, 78 p. http://pubs.er.usgs.gov/publication/ofr20151226
To see what's happening this year at the Comprehensive Sturgeon Research Project, click here!
To return to the Comprehensive Sturgeon Research Project Overview, click here!
To return to River Studies, click here!

