Pathology case of the month - Desert Cottontail
Case History: An adult male desert cottontail (Sylvilagus audubonii) was found dead on a ranch in west Texas, U.S.A. Other rabbits had been reported sick in the area; clinical signs included lethargy, uncoordinated movements, and blood around the nose and mouth.
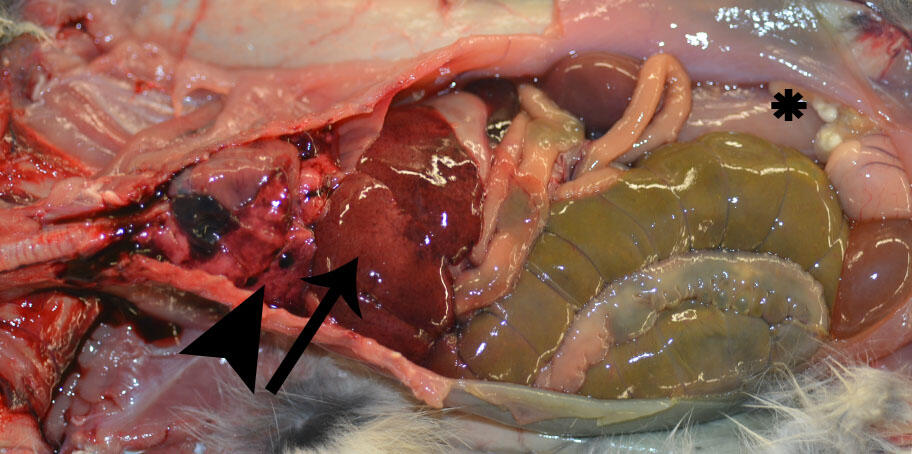
Gross Findings: The rabbit was in poor body condition with depletion of fat stores. There was a small amount of dried blood around the nose and mouth. Lungs were wet and heavy (edema) with multifocal 1-mm to 3-cm diameter dark red areas on the pleural surface (Figure 1; arrowhead); all sections floated in formalin. The liver was moderately friable and mottled dark red to tan with an enhanced reticular pattern and multifocal pinpoint red capsular foci (Figure 1; arrow), as well as a single 1-mm diameter tan focus. The spleen was moderately enlarged (28 x 9 x 5 mm) and dark red. There were intestinal serosal petechia at the ileocecal junction. Throughout the abdomen were multifocal cysts up to 15 x 5 x 2 mm containing clear fluid and white structures (cysticerci) (Figure 1; asterisk). All tissues not described were within normal limits.
Select Histopathological Findings:
Liver: There is massive hepatocellular necrosis with individualization and rounding of hepatocytes (dissociation) (Figure 2A). There is widespread hemorrhage. There is a focal granuloma surrounding necrotic hepatocytes and eosinophilic debris (suspect Taenia sp. migration pathway).
Lung: There is diffuse congestion. There is multifocal mild acute alveolar hemorrhage. Alveoli and bronchioles multifocally contain wispy to homogeneous eosinophilic material and foamy macrophages (edema).
Morphologic Diagnoses:
- Lung: Edema and hemorrhage, subacute, multifocal, moderate
- Liver: Hepatocellular necrosis, massive, acute, marked, with acute to subacute hemorrhage
- Spleen: Congestion, marked
- Spleen: Lymphoid depletion, moderate, with suspect lymphoid necrosis
- Abdominal cysts: Cysticercosis, moderate
- Liver: Granuloma, focal, chronic, mild (suspect Taenia sp. migration)
Condition: Rabbit hemorrhagic disease
Etiology: Family: Caliciviridae; Genus: Lagovirus; Species: Rabbit hemorrhagic disease virus, type 2 (RHDV2); also known as RHDVb and Lagovirus europaeus GI.2
Distribution: Europe, Africa, Australia, New Zealand, and the Americas. First detected in wild North American rabbits and hares in New Mexico in March 2020; as of August 2020, RHDV2 has been detected in wild lagomorphs in New Mexico, Arizona, Colorado, Utah, Nevada, Texas, and California.
Seasonality: Unknown in North America; in Europe, peaks in RHDV2 outbreaks have been associated with the breeding season (Rouco et al. 2017).
Host range: To date, natural infections of RHDV2 have been reported in the European rabbit/domestic rabbit (Oryctolagus cuniculus), desert cottontail (Sylvilagus audubonii), mountain cottontail (S. nuttallii), and some hares of the genus Lepus. Experimental work with RHDV2 has demonstrated morbidity and mortality of infected Eastern cottontails (S. floridanus) (USDA 2018). In contrast, RHDV type 1 is only known to infect wild and domestic European rabbits.
Clinical signs: Free-ranging rabbits and hares are frequently found dead, often with blood around the nose and mouth. Captive animals may also exhibit fever, lethargy/depression, dyspnea, and neurological signs.
Pathogenesis and pathology: Transmission can occur through oral, nasal, or conjunctival exposure. The virus replicates in the liver and can lead to hepatocellular apoptosis, necrosis, liver failure, and disseminated intravascular coagulation. Typical gross findings include hemorrhagic fluid around the nose, a friable, pale tan liver, often with an enhanced reticular pattern (hepatic necrosis), wet lungs with multifocal red areas (edema, congestion, and hemorrhage), and splenomegaly. Disseminated intravascular coagulation can lead to hemorrhage in other tissues such as serosal and epicardial surfaces. Microscopically, common lesions include periportal to panlobular (massive) hepatocellular dissociation and necrosis, hepatocellular degeneration with lipid vacuolar change; splenic congestion, lymphoid depletion, and necrosis; and variable pulmonary edema, congestion, and hemorrhage.
Diagnosis: In the United States, rabbit hemorrhagic disease is a notifiable foreign animal disease and testing for RHDV2 must be performed by USDA’s National Veterinary Services Laboratories (NVSL) or a surrogate laboratory designated by USDA. For wild rabbits, diagnostic testing is primarily directed to either the U.S. Geological Survey National Wildlife Health Center or the Southeastern Cooperative Wildlife Disease Study. Testing options include real-time PCR, ELISA, or antibody testing. Liver is the preferred sample type, and the NVSL real-time PCR is only validated for liver.
Wildlife population impacts: Significant impacts (estimated 60-80% mortality) to susceptible rabbit populations have been reported after introduction of RHDV2 in Australia (Ramsey et al. 2019) and Europe (Delibes-Mateos 2014). Concomitant declines in predator species have also been reported, including to the endangered Iberian lynx (Lynx pardinus) (Delibes-Mateos 2014).
Management: The virus is highly contagious and long-lived in animal tissues and in the environment. Strict biosecurity measures are needed to prevent contact between wild and domestic rabbits, including contact with the secretions or excretions of infected rabbits, contaminated fomites, or mechanical vectors such as flies. To prevent the spread of RHDV2 between populations of wild rabbits, it is recommended that wildlife professionals investigating morbidity or mortality events involving these species wear personal protective equipment (PPE) and decontaminate all gear, clothes, and vehicles. Decontamination protocols include cleaning and disinfecting equipment with 10% bleach, 10% sodium hydroxide, or 1 to 2% formalin.
References:
The Center for Food Security and Public Health, Iowa State University. 2020. Rabbit Hemorrhagic Disease and Other Lagoviruses: Viral Hemorrhagic Disease of Rabbits, Rabbit Calicivirus Disease, European Brown Hare Syndrome, http://www.cfsph.iastate.edu/Factsheets/pdfs/rabbit_hemorrhagic_disease.pdf. Accessed August 2020.
Delibes-Mateos M, Ferreira C, Carro F, Escudero MA, Gortázar, C. 2014. Ecosystem effects of variant rabbit hemorrhagic disease virus, Iberian Peninsula. Emerging infectious diseases 20(12), 2166–2168. https://doi.org/10.3201/eid2012.140517
Joint Pathology Center Systemic Pathology. 2018. Digestive System, D-V31, https://www.askjpc.org/vspo/show_page.php?id=US85Zy9HQ3ZHY3huTHBVSFlHN1FTdz09. Accessed August 2020.
Ramsey DSL, Cox T, Strive T, Forsyth DM, Stuart I, Hall R, Elsworth P, Campbell S. 2019. Emerging RHDV2 suppresses the impact of endemic and novel strains of RHDV on wild rabbit populations. J Appl Ecol 57: 630– 641. https://doi.org/10.1111/1365-2664.13548
Rouco C, Abrantes J, Serronha A, Lopes AM, Maio E, Magahlhaes MJ, Blanco E, Barcena J, Esteves PJ, Santos N, Alves PC, Monterroso P. 2018. Epidemiology of RHDV2 (Lagovirus europaeus/GI.2) in free-living wild European rabbits in Portugal. Transbound Emerg Dis 65(2): e373-e382. doi:10.1111/tbed.12767
United States Department of Agriculture. 2020. USDA APHIS Factsheet: Rabbit Hemorrhagic Disease, https://www.aphis.usda.gov/publications/animal_health/fs-rhdv2.pdf. Accessed August 2020.
United States Department of Agriculture. 2020. USDA Emerging Risk Notice: Rabbit hemorrhagic disease virus, type 2, https://www.aphis.usda.gov/animal_health/downloads/rhdv2.pdf. Accessed August 2020.
USDA APHIS VS NVSL Foreign Animal Disease Diagnostic Laboratory. 2018. Experimental Rabbit Hemorrhagic Disease Virus (RHDV1 and RHDV2) infection in Eastern Cottontail Rabbits. In: Proceedings of the 122nd Annual Meeting of the United States Animal Health Association, United States Animal Health Association, Kansas City, MO, 18-22 October, pp. 59-60.